Impact Factor
Theranostics 2017; 7(17):4340-4349. doi:10.7150/thno.18563 This issue Cite
Research Paper
Sustained Release Talazoparib Implants for Localized Treatment of BRCA1-deficient Breast Cancer
1. Northeastern University, Department of Bioengineering, Boston, MA;
2. Nanomedicine Science and Technology Center, Northeastern University, Boston, MA;
3. Northeastern University, Department of Physics, Boston, MA;
4. Geisel School of Medicine at Dartmouth, Department of Pharmacology, Hanover, NH;
5. Michigan State University, Department of Pharmacology and Toxicology, East Lansing, MI.
Abstract
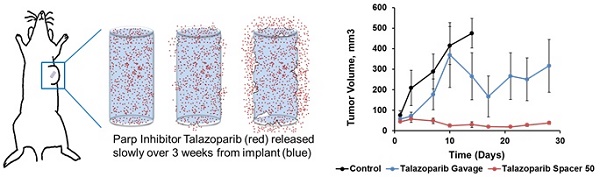
Talazoparib, a potent PARP inhibitor, has shown promising clinical and pre-clinical activity by inducing synthetic lethality in cancers with germline Brca1/2 mutations. Conventional oral delivery of Talazoparib is associated with significant off-target effects, therefore we sought to develop new delivery systems in the form of an implant loaded with Talazoparib for localized, slow and sustained release of the drug at the tumor site in Brca1-deficient breast cancer. Poly(lactic-co-glycolic acid) (PLGA) implants (0.8 mm diameter) loaded with subclinical dose (25 or 50 µg) Talazoparib were fabricated and characterized. In vitro studies with Brca1-deficient W780 and W0069 breast cancer cells were conducted to test sensitivity to PARP inhibition. The in vivo therapeutic efficacy of Talazoparib implants was assessed following a one-time intratumoral injection in Brca1Co/Co;MMTV-Cre;p53+/- mice and compared to drug-free implants and oral gavage. Immunohistochemistry studies were performed on tumor sections using PCNA and γ-H2AX staining. Sustained release of Talazoparib was observed over 28 days in vitro. Mice treated with Talazoparib implants showed statistically significant tumor growth inhibition compared to those receiving drug-free implants or free Talazoparib orally. Talazoparib implants were well-tolerated at both drug doses and resulted in less weight loss than oral gavage. PARP inhibition in mice treated with Talazoparib implants significantly increased double-stranded DNA damage and decreased tumor cell proliferation as shown by PCNA and γ-H2AX staining as compared to controls. These results demonstrate that localized and sustained delivery of Talazoparib via implants has potential to provide superior treatment outcomes at sub-clinical doses with minimal toxicity in patients with BRCA1 deficient tumors.
Keywords: Talazoparib, PARP inhibitor, BRCA-1, breast cancer, local therapy, sustained delivery.
 Global reach, higher impact
Global reach, higher impact