13.3
Impact Factor
Theranostics 2017; 7(18):4359-4369. doi:10.7150/thno.20586 This issue Cite
Research Paper
Clinical Translation and First In-Human Use of [44Sc]Sc-PSMA-617 for PET Imaging of Metastasized Castrate-Resistant Prostate Cancer
1. Department of Nuclear Medicine, University Hospital Bonn, 53127 Bonn, Germany;
2. Institute of Nuclear Chemistry, Johannes Gutenberg University, 55128 Mainz, Germany;
3. Division of Radiopharmaceutical Chemistry, German Cancer Research Center (DKFZ), 69120 Heidelberg, Germany.
* These authors contributed equally
Abstract
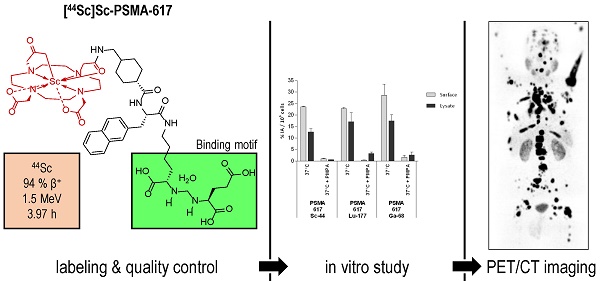
Background: Various trivalent radiometals are well suited for labeling of DOTA-conjugated variants of Glu-ureido-based prostate-specific membrane antigen (PSMA) inhibitors. The DOTA-conjugate PSMA-617 has proven high potential in PSMA radioligand therapy (PSMA-RLT) of prostate cancer as well as PET imaging when labeled with lutetium-177 and gallium-68 respectively. Considering the relatively short physical half-life of gallium-68 this positron emitter precludes prolonged acquisition periods, as required for pre-therapeutic dosimetry or intraoperative applications. In this context, the positron emitter scandium-44 is an attractive alternative for PET imaging. We report the synthesis of [44Sc]Sc-PSMA-617 as radiopharmaceutical with generator produced scandium-44, its in vitro characterization and clinical translation as part of a first in-human study.
Methods: Scandium-44 was obtained from a 44Ti/44Sc radionuclide generator. PSMA-617 was labeled with 142.4±12.7 MBq of scandium-44 in analogy to [68Ga]Ga-PSMA-617 and evaluated in vitro and in cell studies using PSMA+ LNCaP cells. A first-in-human investigation was subsequently carried out in a cohort of 4 patients (mean age 70±1.8 a) registered for [177Lu]Lu-PSMA-617 therapy. 50.5±9.3 MBq (40 µg, 38.4 nmol) [44Sc]Sc-PSMA-617 were applied via intravenous injection (i.v.), respectively. A Siemens Biograph 2 PET/CT system was used to acquire initial dynamic PET data (30 min) of abdomen in list mode followed by static PET/CT data (skull to mid-thigh) at 45 min, 2 and 18 h post-injection (p.i.). For quantitative analysis, dynamic images were reconstructed as 6 data sets of 300 s each. The noise ratio was measured in liver, lung and an additional region outside the body. SUV values in different organs and lesions were measured and compared to [68Ga]Ga-PSMA-11 data of the same patients. Residence times and organ absorbed doses were calculated using OLINDA/EXM software.
Results: Quantitative radiochemical yields of ≥98 % were achieved using 18 nmol of PSMA-617 after 20 min at 95 °C with apparent molar activity of 6.69±0.78 MBq/nmol. Following purification, >99 % radiochemical purity was obtained. [44Sc]Sc-PSMA-617 showed high stability (>95 %) in serum for 24 h. The binding affinity and internalization fraction were determined in PSMA+ LNCaP cells (IC50 = 4.72±0.7 nM and internalization fraction: 15.78±2.14 % IA/106 LNCaP cells) and compared to [68Ga]Ga-PSMA-11 (12.0±2.8 nM and 9.47±2.56 % IA/106 LNCaP cells). Physiological tracer uptake was observed in kidneys, liver, spleen, small intestine, urinary bladder, and salivary glands and pathological uptake in both soft and skeletal metastases. SUV values were significantly lower in the kidneys (14.0) compared to [68Ga]Ga-PSMA-11 OET (30.5). All other measured SUV values did not show a statistically significant difference. Tumor to liver ratios were found to lie between 1.9 and 8.3 for [68Ga]Ga-PSMA-11 and between 2.5 and 8.8 for [44Sc]Sc-PSMA-617 after 120 min. For [44Sc]Sc-PSMA-617 the ratios were higher and no statistically significant differences were observed. Total and % activity were highest in liver followed by kidneys, spleen, small intestine and salivary glands. Rapid wash out was seen in liver and spleen and gradually over time in kidneys. Kidneys received the highest radiation absorbed dose of 0.354 (0.180-0.488) mSv/MBq. No adverse pharmacological effects were observed.
Conclusion: In conclusion [44Sc]Sc-PSMA-617 PET is suitable for PET imaging of prostate cancer tissue. [44Sc]Sc-PSMA-617 shows promise to enable pre-therapeutic dosimetry in clinical settings. However, the clinical advantages for individual dosimetry or other applications like intraoperative applications have to be investigated in further studies.
Keywords: prostate cancer, PSMA-617, scandium-44, PET, theranostics.
 Global reach, higher impact
Global reach, higher impact