13.3
Impact Factor
Theranostics 2018; 8(2):563-574. doi:10.7150/thno.21466 This issue Cite
Research Paper
Local Intratracheal Delivery of Perfluorocarbon Nanoparticles to Lung Cancer Demonstrated with Magnetic Resonance Multimodal Imaging
1. Molecular Imaging Research Center (MIRC), Harbin Medical University, Harbin, Heilongjiang, China 2.TOF-PET/CT/MR center, The Fourth Hospital of Harbin Medical University, Harbin, Heilongjiang, China;
2. Department of Medicine, Division of Cardiology, Washington University School of Medicine, 4320 Forest Park Avenue, Saint Louis, MO 63108, USA.
* The authors contributed equally to this work.
Abstract
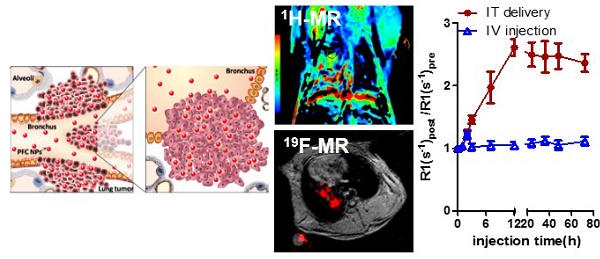
Eighty percent of lung cancers originate as subtle premalignant changes in the airway mucosal epithelial layer of bronchi and alveoli, which evolve and penetrate deeper into the parenchyma. Liquid-ventilation, with perfluorocarbons (PFC) was first demonstrated in rodents in 1966 then subsequently applied as lipid-encapsulated PFC emulsions to improve pulmonary function in neonatal infants suffering with respiratory distress syndrome in 1996. Subsequently, PFC nanoparticles (NP) were extensively studied as intravenous (IV) vascular-constrained nanotechnologies for diagnostic imaging and targeted drug delivery applications.
Methods: This proof-of-concept study compared intratumoral localization of fluorescent paramagnetic (M) PFC NP in the Vx2 rabbit model using proton (1H) and fluorine (19F) magnetic resonance (MR) imaging (3T) following intratracheal (IT) or IV administration. MRI results were corroborated by fluorescence microscopy.
Results: Dynamic 1H-MR and 19F-MR images (3T) obtained over 72 h demonstrated marked and progressive accumulation of M-PFC NP within primary lung Vx2 tumors during the first 12 h post IT administration. Marked 1H and 19F MR signal persisted for over 72 h. In contradistinction, IV M-PFC NP produced a modest transient signal during the initial 2 h post-injection that was consistent circumferential blood pool tumor enhancement. Fluorescence microscopy of excised tumors corroborated the MR results and revealed enormous intratumor NP deposition on day 3 after IT but not IV treatment. Rhodamine-phospholipid incorporated into the PFC nanoparticle surfactant was distributed widely within the tumor on day 3, which is consistent with a hemifusion-based contact drug delivery mechanism previously reported. Fluorescence microscopy also revealed similar high concentrations of M-PFC NP given IT for metastatic Vx2 lung tumors. Biodistribution studies in mice revealed that M-PFC NP given IV distributed into the reticuloendothelial organs, whereas, the same dosage given IT was basically not detected beyond the lung itself. PFC NP given IT did not impact rabbit behavior or impair respiratory function. PFC NP effects on cells in culture were negligible and when given IV or IT no changes in rabbit hematology nor serum clinical chemistry parameters were measured.
Conclusion: IT delivery of PFC NP offered unique opportunity to locally deliver PFC NP in high concentrations into lung cancers with minimal extratumor systemic exposure.
Keywords: perfluorocarbon nanoparticle, MRI, drug delivery, lung cancer, intratracheal delivery.
 Global reach, higher impact
Global reach, higher impact