13.3
Impact Factor
Theranostics 2018; 8(3):610-626. doi:10.7150/thno.22237 This issue Cite
Research Paper
Ligand Activation of PPARγ by Ligustrazine Suppresses Pericyte Functions of Hepatic Stellate Cells via SMRT-Mediated Transrepression of HIF-1α
1. Jiangsu Key Laboratory for Pharmacology and Safety Evaluation of Chinese Materia Medica, Nanjing University of Chinese Medicine, Nanjing 210023, China.
2. Third Institute of Oceanography, State Oceanic Administration, Xiamen 361005, China.
3. Jiangsu Key Laboratory of Therapeutic Material of Chinese Medicine, Nanjing University of Chinese Medicine, Nanjing 210023, China.
4. State Key Laboratory Cultivation Base for TCM Quality and Efficacy, Nanjing University of Chinese Medicine, Nanjing 210023, China.
5. School of Science, China Pharmaceutical University, Nanjing 211198, China.
6. Department of Pathology, School of Medicine, Saint Louis University, MO 63104, USA.
Abstract
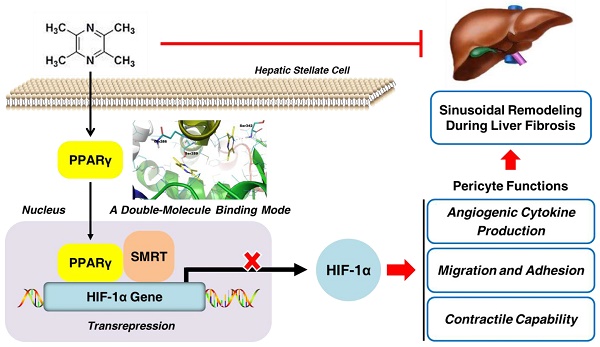
Rationale: Hepatic stellate cells (HSCs) are liver-specific pericytes regulating vascular remodeling during hepatic fibrosis. Here, we investigated how ligustrazine affects HSC pericyte functions.
Methods: Rat HSC-T6 and human HSC-LX2 cells were cultured, and multiple molecular experiments including real-time PCR, Western blot, flow cytometry, immunofluorescence, electrophoretic mobility shift assay and co-immunoprecipitation were used to elucidate the underlying mechanisms. Molecular simulation and site-directed mutagenesis were performed to uncover the target molecule of ligustrazine. Rats were intoxicated with CCl4 for evaluating ligustrazine's effects in vivo.
Results: Ligustrazine inhibited angiogenic cytokine production, migration, adhesion and contraction in HSCs, and activated PPARγ. Selective PPARγ inhibitor GW9662 potently abrogated ligustrazine suppression of HSC pericyte functions. Additionally, HIF-1α inhibitor PX-478 repressed HSC pericyte functions, and ligustrazine inhibited the transcription of HIF-1α, which was diminished by GW9662. Moreover, ligustrazine downregulation of HIF-1α was rescued by knockdown of SMRT, and ligustrazine increased PPARγ physical interaction with SMRT, which was abolished by GW9662. These findings collectively indicated that activation of PPARγ by ligustrazine led to transrepression of HIF-1α via a SMRT-dependent mechanism. Furthermore, molecular docking evidence revealed that ligustrazine bound to PPARγ in a unique double-molecule manner via hydrogen bonding with the residues Ser289 and Ser342. Site-directed mutation of Ser289 and/or Ser342 resulted in the loss of ligustrazine transrepression of HIF-1α in HSCs, indicating that interactions with both the residues were indispensable for ligustrazine effects. Finally, ligustrazine improved hepatic injury, angiogenesis and vascular remodeling in CCl4-induced liver fibrosis in rats.
Conclusions: We discovered a novel ligand activation pattern for PPARγ transrepression of the target gene with therapeutic implications in HSC pericyte biology and liver fibrosis.
Keywords: Hepatic stellate cell, Liver fibrosis, Ligustrazine, PPARγ, HIF-1α, Transrepression, Molecular simulation
 Global reach, higher impact
Global reach, higher impact