13.3
Impact Factor
Theranostics 2018; 8(6):1511-1526. doi:10.7150/thno.21986 This issue Cite
Research Paper
Elevated Expression of miR302-367 in Endothelial Cells Inhibits Developmental Angiogenesis via CDC42/CCND1 Mediated Signaling Pathways
1. Key Laboratory of Arrhythmias of the Ministry of Education of China, Research Center for Translational Medicine, Shanghai East Hospital, Tongji University School of Medicine, Shanghai, 200120, China.
2. Department of Geriatrics, Ruijin Hospital, Jiaotong University, School of Medicine, Shanghai, 200025, China.
3. Department of Medicine and Therapeutics, The Chinese University of Hong Kong, Hong Kong SAR, China
4. Division of Cardiology, Department of Internal Medicine, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan
5. Department of Pharmacology, Center for Translational Medicine, Temple University School of Medicine, Philadelphia, PA 19140, USA.
6. Lab of Cardiovascular Signaling, Centenary Institute, Camperdown, NSW 2050, Australia.
7. Department of Cell and Developmental Biology (R.W., E.E.M.), Department of Medicine (E.E.M.), Penn Cardiovascular Institute (E.E.M.), and Penn Institute for Regenerative Medicine (E.E.M.), University of Pennsylvania, Philadelphia, Pennsylvania 19104, USA.
* These authors contributed equally to this work
Abstract
Rationale: Angiogenesis is critical for embryonic development and microRNAs fine-tune this process, but the underlying mechanisms remain incompletely understood.
Methods: Endothelial cell (EC) specific miR302-367 line was used as gain-of-function and anti-miRs as loss-of-function models to investigate the effects of miR302-367 on developmental angiogenesis with embryonic hindbrain vasculature as an in vivo model and fibrin gel beads and tube formation assay as in vitro models. Cell migration was evaluated by Boyden chamber and scratch wound healing assay and cell proliferation by cell count, MTT assay, Ki67 immunostaining and PI cell cycle analysis. RNA high-throughput sequencing identified miR-target genes confirmed by chromatin immunoprecipitation and 3'-UTR luciferase reporter assay, and finally target site blocker determined the pathway contributing significantly to the phenotype observed upon microRNA expression.
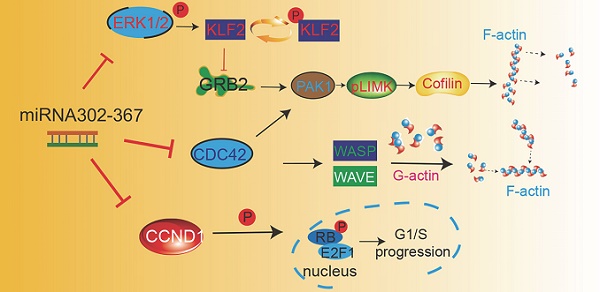
Results: Elevated EC miR302-367 expression reduced developmental angiogenesis, whereas it was enhanced by inhibition of miR302-367, possibly due to the intrinsic inhibitory effects on EC migration and proliferation. We identified Cdc42 as a direct target gene and elevated EC miR302-367 decreased total and active Cdc42, and further inhibited F-actin formation via the WASP and Klf2/Grb2/Pak1/LIM-kinase/Cofilin pathways. MiR302-367-mediated-Klf2 regulation of Grb2 for fine-tuning Pak1 activation contributing to the inhibited F-actin formation, and then the attenuation of EC migration. Moreover, miR302-367 directly down-regulated EC Ccnd1 and impaired cell proliferation via the Rb/E2F pathway.
Conclusion: miR302-367 regulation of endothelial Cdc42 and Ccnd1 signal pathways for EC migration and proliferation advances our understanding of developmental angiogenesis, and meanwhile provides a rationale for future interventions of pathological angiogenesis that shares many common features of physiological angiogenesis.
Keywords: miR302-367, endothelial cells, developmental angiogenesis, cdc42, actin remodeling, cell cycle.
 Global reach, higher impact
Global reach, higher impact