13.3
Impact Factor
Theranostics 2018; 8(6):1527-1539. doi:10.7150/thno.22699 This issue Cite
Research Paper
PRL-3 is a potential glioblastoma prognostic marker and promotes glioblastoma progression by enhancing MMP7 through the ERK and JNK pathways
1. Department of Pathophysiology, School of Basic Medicine, Fourth Military Medical University, Xi'an, China, 710032
2. State Key Laboratory of Cancer Biology, Biotechnology Center, School of Pharmacy, Fourth Military Medical University, Xi'an, China, 710032
3. Experimental Teaching Center of Basic Medicine, Fourth Military Medical University, Xi'an, China, 710032
4. Department of Pathology, Tangdu Hospital, The Fourth Military Medical University, Xi'an, China, 710038
5. Department of General Surgery, Tangdu Hospital, The Fourth Military Medical University, Xi'an, China, 710038
6. Department of Neurosurgery, Xijing Hospital, Fourth Military Medical University, Xi'an, China, 710032
7. Department of Health Statistics, School of Public Health, Fourth Military Medical University, Xi'an, China, 710032
*These authors contributed equally to this work.
Abstract
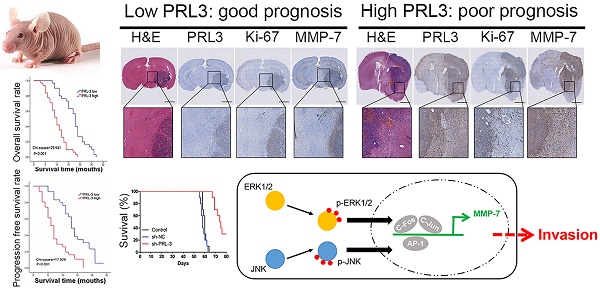
Purpose: Glioblastoma is the most common and aggressive type of primary brain malignancy and is associated with a poor prognosis. Previously, we found that phosphatase of regenerating liver-3 (PRL-3) was significantly up-regulated in glioblastoma as determined by a microarray analysis. However, the function of PRL-3 in glioblastoma remains unknown. We aimed to investigate the clinical relationship between PRL-3 and glioblastoma, and uncover the mechanisms of PRL-3 in the process of glioblastoma.
Methods: PRL-3 expression was evaluated in 61 glioblastoma samples and 4 cell lines by RT-qPCR and immunohistochemistry. Kaplan-Meier analysis was performed to evaluate the prognostic value of PRL-3 for overall survival (OS) and progression-free survival (PFS) for glioblastoma patients. Proliferation was evaluated by Cell Counting Kit-8 (CCK-8) assay and EdU proliferation assay, migration and invasion by wound-closure/Transwell assays, and qRT-PCR/immunoblotting/IHC were used for both in vivo and in vitro investigations.
Result: A high PRL-3 expression level was closely correlated with unfavorable OS and PFS for glioblastoma patients, and was also significantly correlated with Ki-67 expression. Down-regulation of PRL-3 inhibited glioma cell proliferation, invasion and migration through ERK/JNK/matrix metalloproteinase 7 (MMP7) in vitro and in vivo.
Conclusions: PRL-3 expression enhances the invasion and proliferation of glioma cells, highlighting this phosphatase as a novel prognostic candidate and an attractive target for future therapy in glioblastoma.
Keywords: PRL-3, glioblastoma, poor prognosis, prognostic marker
 Global reach, higher impact
Global reach, higher impact