13.3
Impact Factor
Theranostics 2018; 8(13):3571-3583. doi:10.7150/thno.25194 This issue Cite
Research Paper
Transcription factor AP-4 promotes tumorigenic capability and activates the Wnt/β-catenin pathway in hepatocellular carcinoma
1. Guangdong Provincial Key Laboratory of Liver Disease, The Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou 510630, China
2. Guangdong Province Key Laboratory of Brain Function and Disease, Department of Biochemistry, Zhongshan School of Medicine, Sun Yat-sen University, Guangzhou 510080, China
3. Department of Infectious Diseases, Third Affiliated Hospital, Sun Yat-sen University Guangzhou 510630, China
4. Key Laboratory of Protein Modification and Degradation, School of Basic Medical Sciences; Affiliated Cancer Hospital &Institute of Guangzhou Medical University, Guangzhou Medical University, Guangzhou 510030, China
5. State Key Laboratory of Oncology in Southern China, Department of Experimental Research, Sun Yat-sen University Cancer Center, Guangzhou 510060, China
* These authors contributed equally to this work.
Abstract
It has been reported that the transcription factor activating enhancer-binding protein 4 (TFAP4) is upregulated and associated with an aggressive phenotype in several cancers. However, the precise mechanisms underlying the oncogenic role of TFAP4 remain largely unknown.
Methods: TFAP4 expression levels in hepatocellular carcinoma (HCC) cells and tissues were detected by quantitative real-time PCR (qPCR) and immunohistochemistry (IHC). In vitro and in vivo assays were performed to investigate the oncogenic function of TFAP4 in the tumor-initiating cell (TIC)-like phenotype and the tumorigenic capability of HCC cells. Luciferase reporter and chromatin immunoprecipitation (ChIP)-qPCR assays were performed to determine the underlying mechanism of TFAP4-mediated HCC aggressiveness.
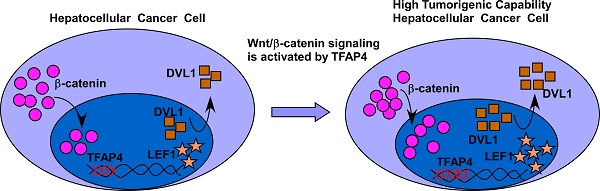
Results: TFAP4 was markedly upregulated in human HCC, and was associated with significantly poorer overall and relapse-free survival in patients with HCC. Furthermore, we found that overexpression of TFAP4 significantly enhanced, whereas silencing TFAP4 inhibited, the tumor sphere formation ability and proportion of side-population cells in HCC cells in vitro, and ectopic TFAP4 enhanced the tumorigenicity of HCC cells in vivo. Mechanistically, we demonstrated that TFAP4 played an important role in activating Wnt/β-catenin signaling by directly binding to the promoters of DVL1 (dishevelled segment polarity protein 1) and LEF1 (lymphoid enhancer binding factor 1).
Conclusions: Our results provide new insight into the mechanisms underlying hyperactivation of the Wnt/β-catenin pathway in HCC, as well the oncogenic ability of TFAP4 to enhance the tumor-forming ability of HCC cells.
Keywords: TFAP4, hepatocellular carcinoma, tumorigenicity, Wnt/β-catenin signaling
 Global reach, higher impact
Global reach, higher impact