13.3
Impact Factor
Theranostics 2018; 8(13):3611-3628. doi:10.7150/thno.25613 This issue Cite
Research Paper
Distinct immune signatures in directly treated and distant tumors result from TLR adjuvants and focal ablation
1. University of California, Davis, Department of Biomedical Engineering, 451 Health Sciences Drive, Davis, CA 95616, USA
2. University of California, Davis, Department of Radiation Oncology, 4501 X Street, Sacramento, CA 95817
3. University of California, Davis, Department of Dermatology, 2921 Stockton Blvd., Institute for Regenerative Cures, Suite 1630, Sacramento, CA 95817, USA
4. University of California, Davis, Center for Comparative Medicine, Davis, CA 95616, USA
5. University of California, Davis School of Medicine, Department of Biochemistry and Molecular Medicine, 4645 Second Avenue, Sacramento, CA 95817, USA
† Authors contributed equally to this work
Abstract
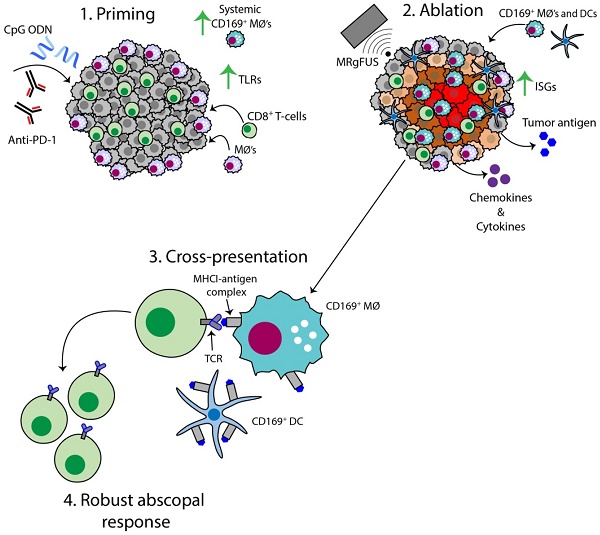
Both adjuvants and focal ablation can alter the local innate immune system and trigger a highly effective systemic response. Our goal is to determine the impact of these treatments on directly treated and distant disease and the mechanisms for the enhanced response obtained by combinatorial treatments.
Methods: We combined RNA-sequencing, flow cytometry and TCR-sequencing to dissect the impact of immunotherapy and of immunotherapy combined with ablation on local and systemic immune components.
Results: With administration of a toll-like receptor agonist agonist (CpG) alone or CpG combined with same-site ablation, we found dramatic differences between the local and distant tumor environments, where the directly treated tumors were skewed to high expression of F4/80, Cd11b and Tnf and the distant tumors to enhanced Cd11c, Cd3 and Ifng. When ablation was added to immunotherapy, 100% (n=20/20) of directly treated tumors and 90% (n=18/20) of distant tumors were responsive. Comparing the combined ablation-immunotherapy treatment to immunotherapy alone, we find three major mechanistic differences. First, while ablation alone enhanced intratumoral antigen cross-presentation (up to ~8% of CD45+ cells), systemic cross-presentation of tumor antigen remained low. Combining same-site ablation with CpG amplified cross-presentation in the draining lymph node (~16% of CD45+ cells) compared to the ablation-only (~0.1% of CD45+ cells) and immunotherapy-only cohorts (~10% of CD45+ cells). Macrophages and DCs process and present this antigen to CD8+ T-cells, increasing the number of unique T-cell receptor rearrangements in distant tumors. Second, type I interferon (IFN) release from tumor cells increased with the ablation-immunotherapy treatment as compared with ablation or immunotherapy alone. Type I IFN release is synergistic with toll-like receptor activation in enhancing cytokine and chemokine expression. Expression of genes associated with T-cell activation and stimulation (Eomes, Prf1 and Icos) was 27, 56 and 89-fold higher with ablation-immunotherapy treatment as compared to the no-treatment controls (and 12, 32 and 60-fold higher for immunotherapy-only treatment as compared to the no-treatment controls). Third, we found that the ablation-immunotherapy treatment polarized macrophages and dendritic cells towards a CD169 subset systemically, where CD169+ macrophages are an IFN-enhanced subpopulation associated with dead-cell antigen presentation.
Conclusion: While the local and distant responses are distinct, CpG combined with ablative focal therapy drives a highly effective systemic immune response.
Keywords: ablation, RNA sequencing, immunotherapy, T-cell receptor sequencing, ultrasound
 Global reach, higher impact
Global reach, higher impact