13.3
Impact Factor
Theranostics 2018; 8(13):3629-3642. doi:10.7150/thno.25955 This issue Cite
Research Paper
Development of a novel peptide aptamer-based immunoassay to detect Zika virus in serum and urine
Zoonosis Research Center, Department of Infection Biology, School of Medicine, Wonkwang University, 460, Iksan-daero, Iksan, 54538, Republic of Korea
‡These authors contributed equally to this manuscript.
Abstract
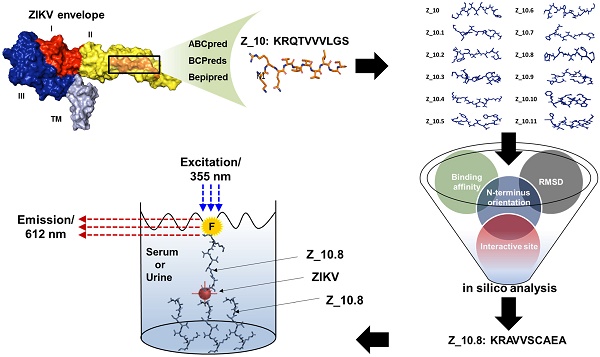
Zika virus (ZIKV) has been identified as a cause of adverse outcomes of pregnancy, including microcephaly and other congenital diseases. Most people infected with ZIKV do not show any symptoms. Development of a method to discriminate dengue virus (DENV) and ZIKV infections has been challenging, and efficient assays for patient management are limited, attributable to high levels of cross-reactivity among co-circulating Flaviviruses. Thus, there is an urgent need for a specific high-throughput diagnostic assay to discriminate ZIKV infections from other Flavivirus infections.
Methods: A novel epitope peptide of the ZIKV envelope protein was predicted using three immune epitope database analysis tools and then further modified. A molecular docking study was conducted using three-dimensional structures of the ZIKV envelope and peptide. Experimentally, interactions between the selected peptides and virus were assessed via a fluorescence-linked sandwich immunosorbent assay (FLISA), and performance of peptide-linked sandwich FLISA was evaluated in virus-spiked human serum and urine.
Results: The Z_10.8 peptide (KRAVVSCAEA) was predicted to be a suitable detector, with a higher binding affinity than other candidates based on four criteria (binding affinity, root mean square deviation, position of amine residue of lysine at the N-terminus, and interactive site) in a docking study. Z_10.8 was significantly more efficient at detecting ZIKV than the other two peptides, as shown in the direct FLISA (P < 0.001). Further, the equilibrium dissociation constant (Kd) for the Z_10.8 peptide was 706.0 ± 177.9 (mean ± SD, nM), with specificity to discriminate ZIKV from DENV. The limit of detection for the sandwich FLISA was calculated as 1×104 tissue culture infective dose (TCID)50/mL. The presence of serum or urine did not interfere with the performance of the Z_10.8-linked sandwich FLISA.
Conclusion: Four criteria are suggested for the development of an in silico modeled peptide aptamer; this computerized peptide aptamer discriminated ZIKV from DENV via immunoassay.
Keywords: Zika virus, peptide aptamer, immunoassay, serum, urine
 Global reach, higher impact
Global reach, higher impact