13.3
Impact Factor
Theranostics 2018; 8(18):5143-5158. doi:10.7150/thno.27310 This issue Cite
Research Paper
Overexpression of HOXC10 promotes angiogenesis in human glioma via interaction with PRMT5 and upregulation of VEGFA expression
1. Program of cancer research, Affiliated Guangzhou Women and Children's Hospital, Department of Biochemistry, Zhongshan School of Medicine, Sun Yat-sen University, China;
2. Guangdong Province Key Laboratory of Brain Function and Disease, Department of Biochemistry, Zhongshan School of Medicine, Sun Yat-sen University, Guangzhou, China;
3. Department of Neurosurgery, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, Guangdong, China;
4. Department of Neurosurgery, the First Affiliated Hospital of Jinan University, Guangzhou, Guangdong, China;
5. Neurosurgical Research Institute, the First Affiliated Hospital of Guangdong Pharmaceutics University, Guangzhou, Guangdong, China;
6. Clinical Experimental Center, Department of Pathology (Clinical Biobanks), Jiangmen Central Hospital, Affiliated Jiangmen Hospital of Sun Yat-sen University, Jiangmen, Guangdong, China;
7. Department of Endocrinology and Diabetes Center, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, Guangdong, China;
8. Department of Pharmacology, Zhongshan School of Medicine, Sun Yat-Sen University, Guangzhou, Guangdong, China.
*These authors contributed equally to this work.
Abstract
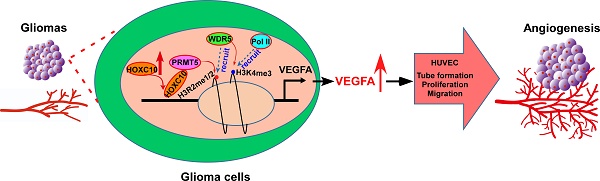
High levels of angiogenesis are associated with poor prognosis in patients with gliomas. However, the molecular mechanisms underlying tumor angiogenesis remain unclear.
Methods: The effect of homeobox C10 (HOXC10) on tube formation, migration, and proliferation of human umbilical vein endothelial cells (HUVECs) and on chicken chorioallantoic membranes (CAMs) was examined. An animal xenograft model was used to examine the effect of HOXC10 on xenograft angiogenesis or the effect of bevacizumab, a monoclonal antibody against vascular endothelial growth factor A (VEGFA), on HOXC10-overexpressing xenografts. A chromatin immunoprecipitation assay was applied to investigate the mechanism in which HOXC10 regulated VEGFA expression.
Results: Overexpressing HOXC10 enhanced the capacity of glioma cells to induce tube formation, migration and proliferation of HUVECs, and neovascularization in CAMs, while silencing HOXC10 had the opposite result. We observed that CD31 staining was significantly increased in tumors formed by HOXC10-overexpressing U251MG cells but reduced in HOXC10-silenced tumors. Mechanistically, HOXC10 could transcriptionally upregulate VEGFA expression by binding to its promoter. Strikingly, treatment with bevacizumab, a monoclonal antibody against VEGFA, significantly inhibited the growth of HOXC10-overexpressing tumors and efficiently impaired angiogenesis. Protein arginine methyltransferase 5 (PRMT5) and WD repeat domain 5 (WDR5), both of which regulate histone post-translational modifications, were required for HOXC10-mediated VEGFA upregulation. Importantly, a significant correlation between HOXC10 levels and VEGFA expression was observed in a cohort of human gliomas.
Conclusions: This study suggests that HOXC10 induces glioma angiogenesis by transcriptionally upregulating VEGFA expression, and may represent a potential target for antiangiogenic therapy in gliomas.
Keywords: HOXC10, glioma, angiogenesis, VEGFA, bevacizumab
 Global reach, higher impact
Global reach, higher impact