13.3
Impact Factor
Theranostics 2018; 8(22):6248-6262. doi:10.7150/thno.28721 This issue Cite
Research Paper
CD44ICD promotes breast cancer stemness via PFKFB4-mediated glucose metabolism
1. School of Medicine, Nankai University, Tianjin 300071, China;
2. The First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, China;
3. Department of Surgery, Chinese PLA General Hospital, Beijing 100071, China;
4. Department of Pathology and Institute of Precision Medicine, Jining Medical University, Jining 272067, China;
5. Department of Medicine, Vanderbilt University Medical Center, Nashville, Tennessee 37212, USA;
6. Department of Reproductive Medicine, School of Medicine and Moores Cancer Center, University of California, San Diego, La Jolla, California 39216, USA;
7. Tianjin Key Laboratory of Tumor Microenvironment and Neurovascular Regulation, Tianjin 300071, China;
8. Project Collaborative Innovation Center for Biotherapy of Ministry of Education 2011, Tianjin 300071, China.
Abstract
CD44 is a single-pass cell surface glycoprotein that is distinguished as the first molecule used to identify cancer stem cells in solid tumors based on its expression. In this regard, the CD44high cell population demonstrates not only the ability to regenerate a heterogeneous tumor, but also the ability to self-regenerate when transplanted into immune-deficient mice. However, the exact role of CD44 in cancer stem cells remains unclear in part because CD44 exists in various isoforms due to alternative splicing.
Methods: Gain- and loss-of-function methods in different models were used to investigate the effects of CD44 on breast cancer stemness. Cancer stemness was analyzed by detecting SOX2, OCT4 and NANOG expression, ALDH activity, side population (SP) and sphere formation. Glucose consumption, lactate secretion and reactive oxygen species (ROS) levels were detected to assess glycolysis. Western blot, immunohistochemical staining, ELISA and TCGA dataset analysis were performed to determine the association of CD44ICD and PFKFB4 with clinical cases. A PFKFB4 inhibitor, 5MPN, was used in a xenograft model to inhibit breast cancer development.
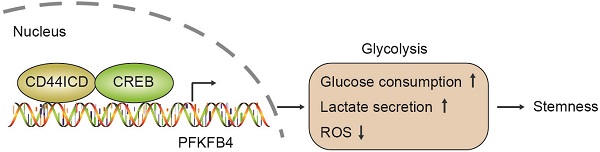
Results: In this report, we found that the shortest CD44 isoform (CD44s) inhibits breast cancer stemness, whereas the cleaved product of CD44 (CD44ICD) promotes breast cancer stemness. Furthermore, CD44ICD interacts with CREB and binds to the promoter region of PFKFB4, thereby regulating PFKFB4 transcription and expression. The resultant PFKFB4 expression facilitates the glycolysis pathway (vis-à-vis oxidative phosphorylation) and promotes stemness of breast cancer. In addition, we found that CD44ICD and PFKFB4 expressions are generally up-regulated in the tumor portion of breast cancer patient samples. Most importantly, we found that 5MPN (a selective inhibitor of PFKFB4) suppresses CD44ICD-induced tumor development.
Conclusion: CD44ICD promotes breast cancer stemness via PFKFB4-mediated glycolysis, and therapies that target PFKFB4 (e.g., 5MPN therapy) may lead to improved outcomes for cancer patients.
Keywords: PFKFB4, CD44ICD, stemness, glucose metabolism
 Global reach, higher impact
Global reach, higher impact