13.3
Impact Factor
Theranostics 2019; 9(4):1144-1153. doi:10.7150/thno.29405 This issue Cite
Research Paper
Synthesis of enantiopure 18F-trifluoromethyl cysteine as a structure-mimetic amino acid tracer for glioma imaging
1. Department of Nuclear Medicine, Guangdong Engineering Research Center for Translational Application of Medical Radiopharmaceuticals, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou 510080, People's Republic of China.
2. The State Key Laboratory of Medicinal Chemical Biology, College of Pharmacy, Nankai University, Tianjin 300350, People's Republic of China.
3. Department of Nuclear Medicine, The Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou 510655, People's Republic of China.
4. College of Materials and Energy, South China Agricultural University, Guangzhou 510642, People's Republic of China.
5. Department of Radiation Oncology, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou 510080, People's Republic of China.
6. School of Pharmaceutical Science and Technology, Tianjin University, Tianjin 300072, People's Republic of China.
*These authors contributed equally.
Abstract
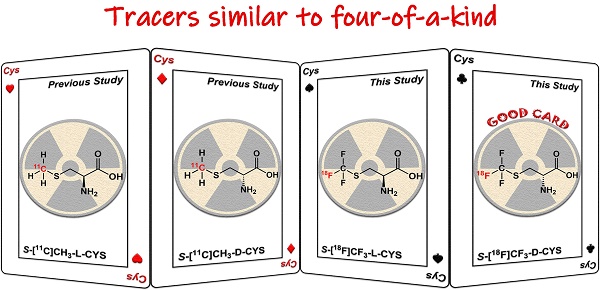
Although 11C-labelled sulfur-containing amino acids (SAAs) including L-methyl-[11C]methionine and S-[11C]-methyl-L-cysteine, are attractive tracers for glioma positron emission tomography (PET) imaging, their applications are limited by the short half-life of the radionuclide 11C (t1/2 = 20.4 min). However, development of 18F-labelled SAAs (18F, t1/2 = 109.8 min) without significant structural changes or relying on prosthetic groups remains to be a great challenge due to the absence of adequate space for chemical modification.
Methods: We herein present 18F-trifluoromethylated D- and L-cysteines which were designed by replacing the methyl group with 18F-trifluoromethyl group using a structure-based bioisosterism strategy. These two enantiomers were synthesized stereoselectively from serine-derived cyclic sulfamidates via a nucleophilic 18F-trifluoromethylthiolation reaction followed by a deprotection reaction. Furthermore, we conducted preliminary in vitro and in vivo studies to investigate the feasibility of using 18F-trifluoromethylated cysteines as PET tracers for glioma imaging.
Results: The two-step radiosynthesis provided the desired products in excellent enantiopurity (ee > 99%) with 14% ± 3% of radiochemical yield. In vitro cell study demonstrated that both enantiomers were taken up efficiently by C6 tumor cells and were mainly transported by systems L and ASC. Among them, the D-enantiomer exhibited relatively good stability and high tumor-specific accumulation in the animal studies.
Conclusion: Our findings indicate that 18F-trifluoromethylated D-cysteine, a new SAA tracer, may be a potential candidate for glioma imaging. Taken together, our study represents a first step toward developing 18F-trifluoromethylated cysteines as structure-mimetic tracers for PET tumor imaging.
Keywords: Positron emission tomography, 18F-trifluoromethylthiolation, 18F-trifluoromethylated cysteine, 18F-labelled sulfur-containing amino acid, glioma imaging
 Global reach, higher impact
Global reach, higher impact