13.3
Impact Factor
Theranostics 2019; 9(10):2868-2881. doi:10.7150/thno.28892 This issue Cite
Research Paper
Imaging fibrosis in inflammatory diseases: targeting the exposed extracellular matrix
1. Werner Siemens Imaging Center, Department of Preclinical Imaging and Radiopharmacy, Eberhard Karls University Tübingen, 72076 Tübingen, Germany
2. Department of Dermatology, University Medical Center, Eberhard Karls University Tübingen, 72076 Tübingen, Germany
3. Department of Women's Health, Research Institute for Women's Health, Eberhard Karls University Tübingen, 72074 Tübingen, Germany
4. The Natural and Medical Sciences Institute (NMI) at the University of Tübingen, Markwiesenstr. 55, 72770 Reutlingen, Germany
5. Department of Medicine/ Cardiology, Cardiovascular Research Laboratories, David Geffen School of Medicine at UCLA, 675 Charles E. Young Drive South, MRL 3645, Los Angeles, CA, USA
6. Institute of Pathology, University Hospital Tübingen, Eberhard Karls University Tübingen, 72076 Tübingen, Germany.
7. Department of Cardiology and Cardiovascular Medicine, University Hospital Tübingen, University of Tübingen, 72076 Tübingen, Germany.
* These authors contributed equally to this work.
Abstract
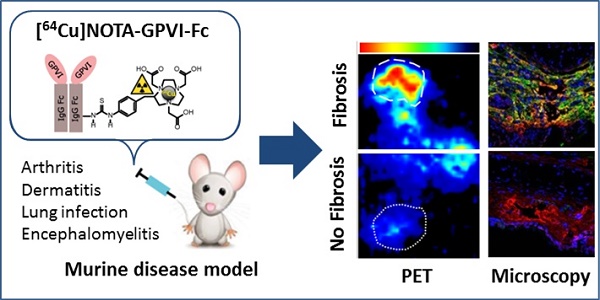
In a variety of diseases, from benign to life-threatening ones, inflammation plays a major role. Monitoring the intensity and extent of a multifaceted inflammatory process has become a cornerstone in diagnostics and therapy monitoring. However, the current tools lack the ability to provide insight into one of its most crucial aspects, namely, the alteration of the extracellular matrix (ECM). Using a radiolabeled platelet glycoprotein VI-based ECM-targeting fusion protein (GPVI-Fc), we investigated how binding of GPVI-Fc on fibrous tissue could uncover the progression of several inflammatory disease models at different stages (rheumatoid arthritis, cutaneous delayed-type hypersensitivity, lung inflammation and experimental autoimmune encephalomyelitis).
Methods: The fusion protein GPVI-Fc was covalently linked to 1,4,7-triazacyclononane-1,4,7-triacetic acid (NOTA) and subsequently labeled with 64Cu. We analyzed noninvasively in vivo 64Cu-GPVI-Fc accumulation in murine cutaneous delayed-type hypersensitivity, anti-glucose-6-phosphate isomerase serum-induced rheumatoid arthritis, lipopolysaccharide-induced lung inflammation and an experimental autoimmune encephalomyelitis model. Static and dynamic Positron Emission Tomography (PET) of the radiotracer distribution was performed in vivo, with ex vivo autoradiography confirmation, yielding quantitative accumulation and a distribution map of 64Cu-GPVI-Fc. Ex vivo tissue histological staining was performed on harvested samples to highlight the fusion protein binding to collagen I, II and III, fibronectin and fibrinogen as well as the morphology of excised tissue.
Results: 64Cu-GPVI-Fc showed a several-fold increased uptake in inflamed tissue compared to control tissue, particularly in the RA model, with a peak 24 h after radiotracer injection of up to half the injected dose. Blocking and isotype control experiments indicated a target-driven accumulation of the radiotracer in the case of chronic inflammation. Histological analysis confirmed a prolonged accumulation at the inflammation site, with a pronounced colocalization with the different components of the ECM (collagen III and fibronectin notably). Binding of the fusion protein appeared to be specific to the ECM but unspecific to particular components.
Conclusion: Imaging of 64Cu-GPVI-Fc accumulation in the ECM matrix appears to be a promising candidate for monitoring chronic inflammation. By binding to exposed fibrous tissue (collagen, fibronectin, etc.) after extravasation, a new insight is provided into the fibrotic events resulting from a prolonged inflammatory state.
Keywords: fibrosis, positron emission tomography, glycoprotein VI
 Global reach, higher impact
Global reach, higher impact