13.3
Impact Factor
Theranostics 2019; 9(15):4474-4493. doi:10.7150/thno.31942 This issue Cite
Research Paper
Astrocytic connexin 43 potentiates myelin injury in ischemic white matter disease
1. Department of Neurology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China.
2. State Key Laboratory of Magnetic Resonance and Atomic and Molecular Physics, Key Laboratory of Magnetic Resonance in Biological Systems, Wuhan Institute of Physics and Mathematics, Chinese Academy of Science, Wuhan 430030, China.
3. Center for Translational Neuromedicine, University of Rochester Medical Center, Rochester, NY 14642, USA.
4. Center for Neuroscience, University of Copenhagen Faculty of Health and Medicine, Copenhagen, Denmark.
5. Center for Neuroscience, Rigshospitalet, Copenhagen, Denmark.
6. Key Laboratory of Neurological Diseases of Chinese Ministry of Education, the School of Basic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China.
*Drs. Minghuan Wang, Chuan Qin and Xiang Luo contribute equally to this work.
Abstract
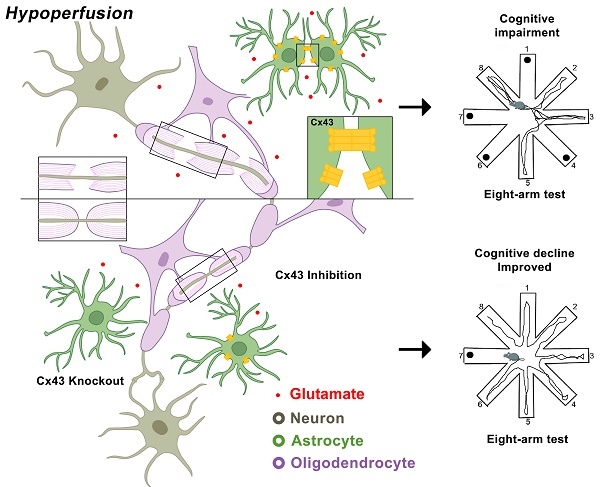
Rational: Myelin loss is a characteristic feature of both ischemic white matter disease and its associated vascular dementia, and is a hallmark of chronic cerebral hypoperfusion due to carotid artery stenosis. Yet the cellular mechanisms involved in ischemic dysmyelination are not well-understood, and no effective treatment has emerged to prevent or slow hypoperfusion-related demyelination. In a study employing the bilateral common carotid artery stenosis (BCAS) mouse model, we found reduced cerebral blood flow velocity and arteriolar pulsatility, and confirmed that prolonged BCAS provoked myelin disruption. These pathological features were associated with marked cognitive decline, in the absence of evident damage to axons.
Methods: To assess the role of astroglial communication in BCAS-associated demyelination, we investigated the effect of deleting or inhibiting connexin 43 (Cx43), a constituent of astroglial gap junctions and hemichannels.
Results: Genetic deletion and pharmacological inhibition of gap junctions both protected myelin integrity and rescued cognitive decline in the BCAS-treated mice. Gap junction inhibition also suppressed the transient increase in extracellular glutamate observed in the callosal white matter of wild-type mice exposed to BCAS.
Conclusion: These findings suggest that astrocytic Cx43 may be a viable target for attenuating the demyelination and cognitive decline associated with chronic cerebral hypoperfusion.
Keywords: connexin 43, myelin injury, ischemic white matter disease, astrocyte
 Global reach, higher impact
Global reach, higher impact