13.3
Impact Factor
Theranostics 2019; 9(22):6396-6411. doi:10.7150/thno.34801 This issue Cite
Research Paper
Macrophages facilitate post myocardial infarction arrhythmias: roles of gap junction and KCa3.1
1. Department of Cardiology, XinHua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China
2. Department of Ophthalmology, XinHua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China
# These authors contributed equally.
Abstract
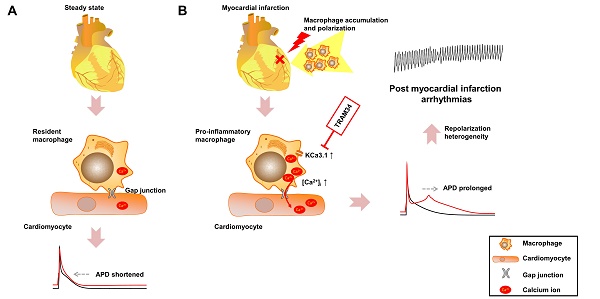
Effective therapeutic targets against post-myocardial infarction (MI) arrhythmias remain to be discovered. We aimed to investigate the role of macrophages in post-MI arrhythmias.
Methods: Mononuclear cell accumulation, macrophage polarization from M0 to M1 subset, and gap junction formation were analyzed in MI patients and MI mice by flow cytometry, immunofluorescence and patch clamping. Differentially expressed genes were identified by RNA sequencing. Macrophages and cardiomyocytes were cocultured in vitro, and the effects of gap junction and KCa3.1 on electrophysiological properties were assessed by patch clamping. The effects of KCa3.1 inhibition on post-MI arrhythmias were assessed by intracardiac stimulation and ambulatory electrocardiograms in vivo.
Results: Percentage of pro-inflammatory mononuclear cells were significantly elevated in patients with post-MI arrhythmias compared with MI patients without arrhythmias and healthy controls (p<0.001). Macrophages formed gap junction with cardiomyocytes in MI border zones of MI patient and mice, and pro-inflammatory macrophages were significantly increased 3 days post-MI (p<0.001). RNA sequencing identified Kcnn4 as the most differentially expressed gene encoding ion channel, and the upregulation is mainly attributed to macrophage accumulation and polarization into pro-inflammatory subset. In vitro coculture experiments demonstrated that connection with M0 macrophages via gap junction slightly shortened the action potential durations (APDs) of cardiomyocytes. However, the APD90 of cardiomyocytes connected with M1 macrophages were significantly prolonged (p<0.001), which were effectively attenuated by gap junction inhibition (p=0.002), KCa3.1 inhibition (p=0.008), KCa3.1 silencing (p<0.001) and store-operated Ca2+ channel inhibition (p=0.005). In vivo results demonstrated that KCa3.1 inhibition significantly decreased the QTc durations (p=0.031), intracardiac stimulation-induced ventricular arrhythmia durations (p=0.050) and incidence of premature ventricular contractions (p=0.030) in MI mice.
Conclusion: Macrophage polarization leads to APD heterogeneity and post-MI arrhythmias via gap junction and KCa3.1 activation. The results provide evidences of a novel mechanism of post-MI heterogeneous repolarization and arrhythmias, rendering macrophages and KCa3.1 to be potential therapeutic targets.
Keywords: myocardial infarction, arrhythmias, macrophage, gap junction, KCa3.1
 Global reach, higher impact
Global reach, higher impact