13.3
Impact Factor
Theranostics 2019; 9(24):7282-7297. doi:10.7150/thno.33920 This issue Cite
Research Paper
LncRNA-Safe contributes to cardiac fibrosis through Safe-Sfrp2-HuR complex in mouse myocardial infarction
1. Department of Cardiovascular Surgery of the First Affiliated Hospital & Institute for Cardiovascular Science, State Key Laboratory of Radiation Medicine and Protection, Medical College, Soochow University, Suzhou 215000, China
2. Fujian Provincial Key Laboratory of Reproductive Health Research, School of Medicine, Xiamen University, Xiamen 361102, China
3. iHuman Institute, School of Life Science and Technology, ShanghaiTech University, Shanghai 210021, China
# Co-first authors
Abstract
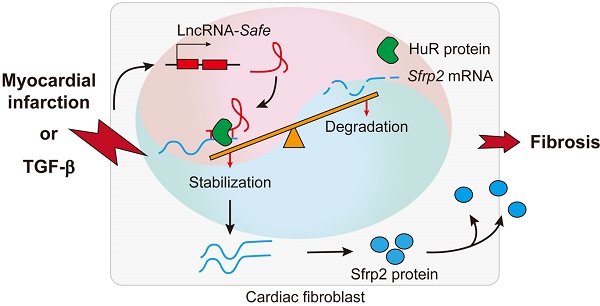
Rationale: As a hallmark of various heart diseases, cardiac fibrosis ultimately leads to end-stage heart failure. Anti-fibrosis is a potential therapeutic strategy for heart failure. Long noncoding RNAs (lncRNAs) have emerged as critical regulators of heart diseases that promise to serve as therapeutic targets. However, few lncRNAs have been directly implicated in cardiac fibrosis.
Methods: The lncRNA expression profiles were assessed by microarray in cardiac fibrotic and remote ventricular tissues in mice with myocardial infarction. The mechanisms and functional significance of lncRNA-AK137033 in cardiac fibrosis were further investigated with both in vitro and in vivo models.
Results: We identified 389 differentially expressed lncRNAs in cardiac fibrotic and remote ventricular tissues in mice with myocardial infarction. Among them, a lncRNA (AK137033) we named Safe was enriched in the nuclei of fibroblasts, and elevated in both myocardial infarction and TGF-β-induced cardiac fibrosis. Knockdown of Safe prevented TGF-β-induced fibroblast-myofibroblast transition, aberrant cell proliferation and secretion of extracellular matrix proteins in vitro, and mended the impaired cardiac function in mice suffering myocardial infarction. In vitro studies indicated that knockdown of Safe significantly inhibited the expression of its neighboring gene Sfrp2, and vice versa. The Sfrp2 overexpression obviously disturbed the regulatory effects of Safe shRNAs in both the in vitro cultured cardiac fibroblasts and myocardial infarction-induced fibrosis. Dual-Luciferase assay demonstrated that Safe and Sfrp2 mRNA stabilized each other via their complementary binding at the 3'-end. RNA electrophoretic mobility shift assay and RNA immunoprecipitation assay indicated that RNA binding protein HuR could bind to Safe-Sfrp2 RNA duplex, whereas the knockdown of HuR dramatically reduced the stabilization of Safe and Sfrp2 mRNAs, down-regulated their expression in cardiac fibroblasts, and thus inhibited TGF-β-induced fibrosis. The Safe overexpression partially restrained the phenotype change of cardiac fibroblasts induced by Sfrp2 shRNAs, but not that induced by HuR shRNAs.
Conclusions: Our study identifies Safe as a critical regulator of cardiac fibrosis, and demonstrates Safe-Sfrp2-HuR complex-mediated Sfrp2 mRNA stability is the underlying mechanism of Safe-regulated cardiac fibrosis. Fibroblast-enriched Safe could represent a novel target for anti-fibrotic therapy in heart diseases.
Keywords: non-coding RNA, fibrosis, cardiac remodeling
 Global reach, higher impact
Global reach, higher impact