13.3
Impact Factor
Theranostics 2020; 10(8):3652-3667. doi:10.7150/thno.41305 This issue Cite
Review
Synthetic immunity by remote control
1. The Wallace H. Coulter Department of Biomedical Engineering, Georgia Institute of Technology & Emory University, Atlanta, GA 30332, USA
2. Institute for Electronics and Nanotechnology, Georgia Institute of Technology, Atlanta, GA 30332, USA
3. Parker H. Petit Institute of Bioengineering and Bioscience, Georgia Institute of Technology, Atlanta, GA 30332, USA
4. Integrated Cancer Research Center, Georgia Institute of Technology, Atlanta, GA 30332, USA
5. Georgia Immunoengineering Consortium, Emory University and Georgia Institute of Technology, Atlanta, GA 30332, USA
*Equal contributions.
Abstract
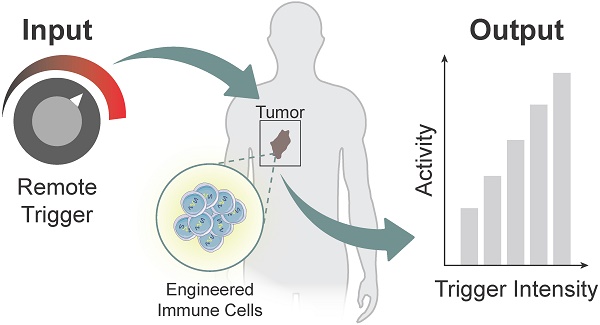
Cell-based immunotherapies, such as T cells engineered with chimeric antigen receptors (CARs), have the potential to cure patients of disease otherwise refractory to conventional treatments. Early-on-treatment and long-term durability of patient responses depend critically on the ability to control the potency of adoptively transferred T cells, as overactivation can lead to complications like cytokine release syndrome, and immunosuppression can result in ineffective responses to therapy. Drugs or biologics (e.g., cytokines) that modulate immune activity are limited by mass transport barriers that reduce the local effective drug concentration, and lack site or target cell specificity that results in toxicity. Emerging technologies that enable site-targeted, remote control of key T cell functions - including proliferation, antigen-sensing, and target-cell killing - have the potential to increase treatment precision and safety profile. These technologies are broadly applicable to other immune cells to expand immune cell therapies across many cancers and diseases. In this review, we highlight the opportunities, challenges and the current state-of-the-art for remote control of synthetic immunity.
Keywords: remote control, synthetic immunity, engineered cells, gene switches, immunotherapy
 Global reach, higher impact
Global reach, higher impact