13.3
Impact Factor
Theranostics 2014; 4(2):163-174. doi:10.7150/thno.7064 This issue Cite
Research Paper
Gold Nanocage-Photosensitizer Conjugates for Dual-Modal Image-Guided Enhanced Photodynamic Therapy
1. Photodynamic Therapy Center, Roswell Park Cancer Institute Buffalo, NY 14263 (USA);
2. Department of Chemistry and Biochemistry, University of Arkansas, Fayetteville, AR 72701 (USA);
3. Department of Biomedical Engineering, The State University of New York, Buffalo, NY 14260 (USA).
4. Departments of Electrical Engineering and Creative IT Engineering, Pohang University of Science and Technology, Pohang 790-784 (Republic of Korea).
5. Department of Biostatistics, Brown University, Providence, RI 02912 (USA).
* These authors equally contributed on this work.
Abstract
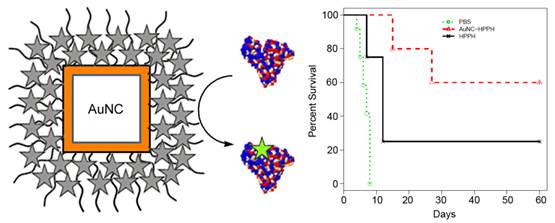
We have demonstrated that gold nanocage-photosensitizer conjugates can enable dual image-guided delivery of photosensitizer and significantly improve the efficacy of photodynamic therapy in a murine model. The photosensitizer, 3-devinyl-3-(1'-hexyloxyethyl)pyropheophorbide (HPPH), was noncovalently entrapped in the poly(ethylene glycol) monolayer coated on the surface of gold nanocages. The conjugate is stable in saline solutions, while incubation in protein rich solutions leads to gradual unloading of the HPPH, which can be monitored optically by fluorescence and photoacoustic imaging. The slow nature of the release in turn results in an increase in accumulation of the drug within implanted tumors due to the passive delivery of gold nanocages. Furthermore, the conjugate is found to generate more therapeutic singlet oxygen and have a lower IC50 value than the free drug alone. Thus the conjugate shows significant suppression of tumor growth as compared to the free drug in vivo. Short-term study showed neither toxicity nor phenotypical changes in mice at therapeutic dose of the conjugates or even at 100-fold higher than therapeutic dose of gold nanocages.
Keywords: Gold Nanostructures, Drug Delivery, Fluorescence Imaging, Photoacoustic Imaging, Cancer Treatment.
 Global reach, higher impact
Global reach, higher impact