13.3
Impact Factor
Theranostics 2017; 7(5):1204-1213. doi:10.7150/thno.17069 This issue Cite
Research Paper
Aptamer Internalization via Endocytosis Inducing S-Phase Arrest and Priming Maver-1 Lymphoma Cells for Cytarabine Chemotherapy
1. Department of Oncology, Xiangya Hospital, Central South University, Changsha, Hunan Province, China.
2. Department of Pathology and Genomic Medicine, Houston Methodist Hospital, Houston, TX, USA.
3. Department of Hematology, Xiangya Hospital, Central South University, Changsha, Hunan Province, China.
4. Department of Hepatology, The First Hospital of Jilin University, Changchun, Jilin Province, China.
5. Department of Nanomedicine, Houston Methodist Research Institute, Houston, TX, USA.
Abstract
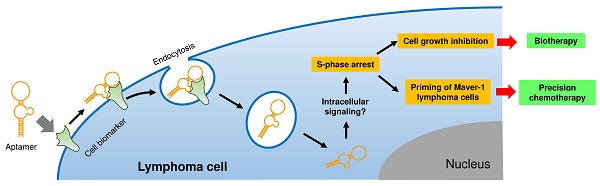
The goal of precision therapy is to efficiently treat cancer without side effects. Aptamers are a class of small ligands composed of single-stranded oligonucleotides that bind to their targets with high affinity and specificity. In this study, we identified an ssDNA aptamer specifically targeting Maver-1 lymphoma cells with high binding affinity (Kd = 70±8 pmol/L). Interestingly, cellular cycle studies revealed that exposure of Maver-1 cells to synthetic aptamers triggered S-phase arrest of 40% of the cells (vs. 18% baseline). Confocal microscopy confirmed specific cell binding of aptamers and the resultant endocytosis into Maver-1 cells. Subsequent functional assays validated the fact that aptamer internalization into targeted cells is a prerequisite for Maver-1 cell growth inhibition. Importantly, aptamer-induced S-phase arrest induced enhanced chemotherapeutic results involving cytarabine, which primarily kills lymphoma cells at S-phase. Combination treatments revealed that aptamer re-exposure considerably primed Maver-1 cells for cytarabine chemotherapy, thus achieving a synergistic killing effect by reaching cell death rates as high as 61% (vs. 13% or 14% induced by aptamer or cytarabine treatment alone). These findings demonstrated that aptamers do not only act as molecular ligands but can also function as biotherapeutic agents by inducing S-phase arrest of lymphoma cells. In addition, logical combination of aptamer and cytarabine treatments ushers the way to a unique approach in precision lymphoma chemotherapy.
Keywords: aptamer, biotherapeutics, lymphoma, synergistic chemotherapy, S-phase arrest
 Global reach, higher impact
Global reach, higher impact