Impact Factor
Theranostics 2012; 2(2):127-138. doi:10.7150/thno.3476 This issue Cite
Review
Analysis of Protease Activity Using Quantum Dots and Resonance Energy Transfer
Department of Life Science, Hanyang University, Seoul 133-791, Republic of Korea
Abstract
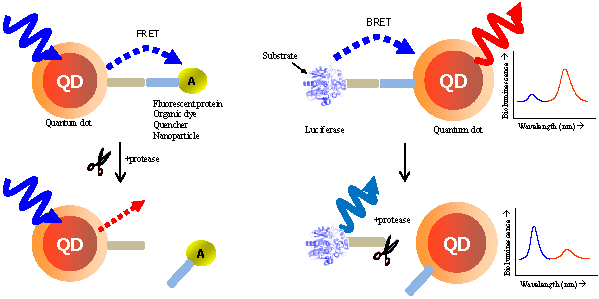
This review demonstrates the detection of protease activity based on the energy transfer of quantum dots (QDs). By incorporation of varying protease substrates into designed QD probes both in fluorescence resonance energy transfer (FRET) and bioluminescence resonance energy transfer (BRET) system, proteolytic activity led to changes in the energy transfer efficiency. Especially due to the superior properties of QDs, it can be served as an excellent probe for a multiplexed and high-throughput protease assay with high sensitivity. It is anticipated that the QD-based FRET/BRET probes will have a great potential for dissecting the fundamental roles of proteases and designing potential protease inhibitors as therapeutic drugs in biology and nanomedicine.
Keywords: protease, quantum dot, energy transfer, BRET, FRET, multiplex, nanoparticle, luciferase, quencher
 Global reach, higher impact
Global reach, higher impact