13.3
Impact Factor
Theranostics 2013; 3(9):703-718. doi:10.7150/thno.5923 This issue Cite
Research Paper
Comparative Tumor Imaging and PDT Efficacy of HPPH Conjugated in the Mono- and Di-Forms to Various Polymethine Cyanine Dyes: Part - 2
1. Photodynamic Therapy Center, Molecular and Cellular Biophysics and Biochemistry, Cell Stress Biology, Roswell Park Cancer Institute, Buffalo, NY 14263;
2. Institute for Lasers, Photonics and Biophotonics, University at Buffalo, Buffalo, NY 14221, USA.
Abstract
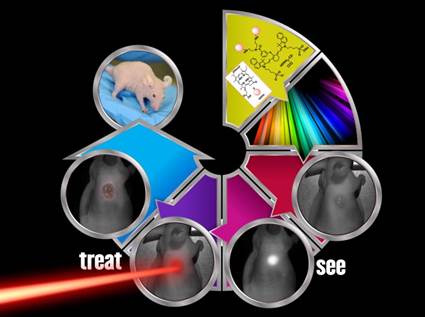
Previous reports from our laboratory have shown that a bifunctional agent obtained by conjugating a photosensitizer (HPPH) to a cyanine dye (CD) can be used for fluorescence image-guided treatment of tumor by photodynamic therapy (PDT). However, the resulting HPPH-CD conjugate showed a significant difference between the tumor-imaging and therapeutic doses. It was demonstrated that the singlet oxygen (1O2*, a key cytotoxic agent in PDT) produced by the conjugate upon excitation of the HPPH moiety was partially quenched by the CD-moiety; this resulted in a reduced PDT response when compared to HPPH-PDT under similar treatment parameters. To improve the therapeutic potential of the conjugate, we synthesized a series of dual functional agents in which one or two HPPH moieties were separately conjugated to three different dyes (Cypate, modified IR820 or modified IR783). The newly synthesized conjugates were compared with our lead compound HPPH-CD in terms of photophysical properties, in vitro and in vivo PDT efficacy, tumor uptake and imaging potential. Among the analogs investigated, the conjugate, in which two HPPH moieties were linked to the modified IR820 produced enhanced tumor uptake and tumor contrast in both Colon 26 (a murine Colon carcinoma) and U87 (a human glioblastoma) cell lines. The long-term PDT efficacy (cure) of this conjugate in BALB/c mice, bearing Colon 26 tumors was also enhanced; however, its efficacy in Nude mice bearing U87 tumors was slightly reduced. It was also found that in all the conjugates the singlet oxygen generation and, consequently, PDT efficacy were compromised by a competing pathway, whereby an electronic excitation of HPPH, the energy donor, is deactivated through an electronic excitation energy transfer (Forster Resonance Energy Transfer, FRET) to the CD fluorophore, the energy acceptor, resulting in overall reduction of the singlet oxygen production. Conjugates with increased FRET showed reduced singlet oxygen production and PDT efficacy. Among the conjugates investigated, the bifunctional agent in which two HPPH moieties were linked to the benzoindole-based cyanine dye 11 showed superiority over the lead candidate 9 (mono HPPH-cyanine dye).
Keywords: Photodynamic therapy, Fluorophores, Reactive Oxygen species, Imaging. Forster Resonance Energy Transfer.
 Global reach, higher impact
Global reach, higher impact