Impact Factor
Theranostics 2015; 5(1):86-96. doi:10.7150/thno.10062 This issue Cite
Research Paper
Codon-optimized Human Sodium Iodide Symporter (opt-hNIS) as a Sensitive Reporter and Efficient Therapeutic Gene
1. Department of Nuclear Medicine, Seoul National University College of Medicine;
2. Biomedical Sciences, Seoul National University College of Medicine;
3. Cancer Research Institute, Seoul National University College of Medicine,
4. Tumor Microenvironment Global Core Research Center, Seoul National University;
5. Molecular Medicine and Biopharmaceutical Sciences, Seoul National University;
6. Cancer Imaging Center, Seoul National University Hospital, Seoul, South Korea;
7. Division of High-Risk Pathogen Research, Korea National Institute of Health, Osong, South Korea.
#These authors are equally contributed in this research.
Abstract
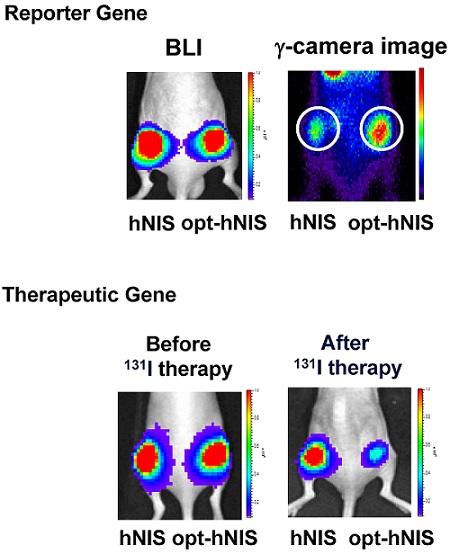
To generate a more efficient in vivo reporter and therapeutic gene, we optimized the coding sequence of the human sodium/iodide symporter (NIS) gene by replacing NIS DNA codons from wild type to new codons having the highest usage in human gene translation. The Codon Adaptation Index (CAI), representing the number of codons effective for human expression, was much improved (0.79 for hNIS, 0.97 for opt-hNIS). Both wild-type (hNIS) and optimized human NIS (opt-hNIS) were cloned into pcDNA3.1 and pMSCV vectors for transfection. Various cancer cell lines such as thyroid (TPC-1, FRO, B-CPAP), breast (MDA-MB-231), liver (Hep3B), cervical (HeLa), and glioma (U87MG) were transfected with pcDNA3.1/hNIS or pcDNA3.1/opt-hNIS. 125I uptake by opt-hNIS-expressing cells was 1.6 ~ 2.1 times higher than uptake by wild-type hNIS-expressing cells. Stable cell lines were also established by retroviral transduction using pMSCV/hNIS or pMSCV/opt-hNIS, revealing higher NIS protein levels and 125I uptake in opt-hNIS-expressing cells than in hNIS-expressing cells. Moreover, scintigraphic images from cell plates and mouse xenografts showed stronger signals from opt-hNIS-expressing cells than hNIS-expressing cells, and radioactivity uptake by opt-hNIS-expressing tumors was 2.3-fold greater than that by hNIS-expressing tumors. To test the efficacy of radioiodine therapy, mouse xenograft models were established with cancer cells expressing hNIS or opt-hNIS. 131I treatment reduced tumor sizes of hNIS- and opt-hNIS-expressing tumors to 0.57- and 0.27- fold, respectively, compared to their sizes before therapy, suggesting an improved therapeutic effect of opt-hNIS. In summary, this study shows that codon optimization strongly increases hNIS protein levels and radioiodine uptake, thus supporting opt-hNIS as a more sensitive reporter and efficient therapeutic gene.
Keywords: human sodium iodide symporter (hNIS), radioiodine therapy, codon optimization, reporter, gene therapy.
 Global reach, higher impact
Global reach, higher impact