Impact Factor
Theranostics 2015; 5(7):746-754. doi:10.7150/thno.10732 This issue Cite
Research Paper
Effect of PEG Pairing on the Efficiency of Cancer-Targeting Liposomes
1. KAIST Institute for the BioCentury, Department of Biological Sciences, Korea Advanced Institute of Science and technology (KAIST), 291 Daehak-ro, Daejeon 305-701, Republic of Korea.
2. Laboratory of Nanomedicine and Biomaterials, Department of Anesthesiology, Brigham and Women's Hospital Harvard Medical School, Boston, MA 02115, U.S.A.
3. School of Life Sciences, Gwangju Institute of Science and Technology, 123-Cheomdangwagi-ro, Gwangju 500-712, Republic of Korea.
Abstract
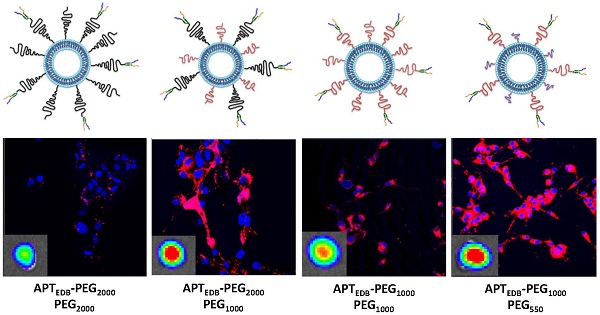
Standardized poly(ethylene glycol)-modified (PEGylated) liposomes, which have been widely used in research as well as in pre-clinical and clinical studies, are typically constructed using PEG with a molecular weight of 2000 Da (PEG2000). Targeting ligands are also generally conjugated using various functionalized PEG2000. However, although standardized protocols have routinely used PEG2000, it is not because this molecular weight PEG has been optimized to enhance tumor uptake of nanoparticles. Herein, we investigated the effect of various PEG lipid pairings—that is, PEG lipids for targeting-ligand conjugation and PEG lipids for achieving 'stealth' function—on in vitro cancer cell- and in vivo tumor-targeting efficacy. A class of high-affinity peptides (aptides) specific to extra domain B of fibronectin (APTEDB) was used as a representative model for a cancer-targeting ligand. We synthesized a set of aptide-conjugated PEGylated phospholipids (APTEDB‑PEG2000‑DSPE and APTEDB‑PEG1000‑DSPE) and then paired them with methoxy-capped PEGylated phospholipids with diverse molecular weights (PEG2000, PEG1000, PEG550, and PEG350) to construct various aptide-conjugated PEGylated liposomes. The liposomes with APTEDB‑PEG2000/PEG1000 and APTEDB‑PEG1000/PEG550 pairings had the highest uptake in EDB-positive cancer cells. Furthermore, in a U87MG xenograft model, APTEDB‑PEG2000/PEG1000 liposomes retarded tumor growth to the greatest extent, followed closely by APTEDB‑PEG1000/PEG550 liposomes. Among the PEGylated liposomes tested, pairs in which the methoxy-capped PEG length was about half that of the targeting ligand-displaying PEG exhibited the best performance, suggesting that PEG pairing is a key consideration in the design of drug-delivery vehicles.
Keywords: aptides · liposomes · cancer therapy · poly(ethylene glycol) · drug delivery · extra domain B of fibronectin
 Global reach, higher impact
Global reach, higher impact