Impact Factor
Theranostics 2015; 5(10):1058-1067. doi:10.7150/thno.11758 This issue Cite
Research Paper
Enzyme-Controlled Intracellular Self-Assembly of 18F Nanoparticles for Enhanced MicroPET Imaging of Tumor
1. School of Chemical and Material Engineering, Jiangnan University, 1800 Lihu Road, Wuxi, Jiangsu 214122, China
2. CAS Key Laboratory of Soft Matter Chemistry, Department of Chemistry, University of Science and Technology of China, Hefei, Anhui 230026, China
3. Key Laboratory of Nuclear Medicine, Ministry of Health, Jiangsu Key Laboratory of Molecular Nuclear Medicine, Jiangsu Institute of Nuclear Medicine, Wuxi, Jiangsu 214063, China
4. College of Pharmaceutical Sciences, Soochow University, Suzhou, Jiangsu 215123, China
‡These authors contributed equally.
Abstract
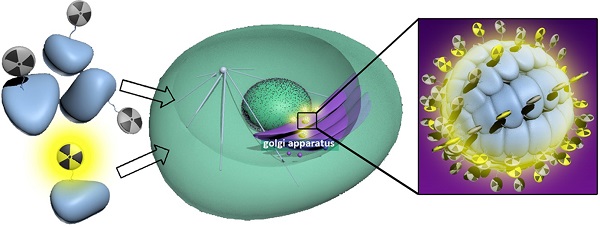
Herein, we report the development of a new “smart” radioactive probe (i.e., 1) which can undergo furin-controlled condensation and self-assembly of radioactive nanoparticles (i.e., 1-NPs) in tumor cells and its application for enhanced microPET imaging of tumors in nude mice co-injected with its cold analog (i.e., 1-Cold). Furin-controlled condensation of 1-Cold and self-assembly of its nanoparticles (i.e., 1-Cold-NPs) in vitro were validated and characterized with HPLC, mass spectra, SEM, and TEM analyses. Cell uptake studies showed that both 1 and 1-Cold have good cell permeability. TEM images of 1-Cold-treated MDA-MB-468 cells directly uncovered that the intracellular 1-Cold-NPs were at/near the location of furin (i.e., Golgi bodies). MTT results indicated that 50 µM 1-Cold did not impose cytotoxicity to MDA-MB-468 cells up to 12 hours. MicroPET imaging of MDA-MB-468 tumor-bearing mice indicated that mice co-injected with 1 and 1-Cold showed higher uptake and longer attenuation of the radioactivity in tumors than those mice only injected with same dosage of 1. Tumor uptake ratios of 1 between these two groups of mice reached the maximum of 8.2 folds at 240 min post injection. Biodistribution study indicated that the uptake ratios of 1 in kidneys between these two groups continuously increased and reached 81.9 folds at 240 min post injection, suggesting the formation of radioactive NPs (i.e., 1-NPs) in MDA-MB-468 tumors of mice co-injected with 1 and 1-Cold. And the nanoparticles were slowly digested and secreted from the tumors, accumulating in the kidneys. Our ''smart'' probe (i.e., 1), together with the strategy of co-injection, might help researchers trace the biomarkers of interest within a longer time window.
Keywords: self-assembly, nanoparticles, furin, co-injection.
 Global reach, higher impact
Global reach, higher impact