Impact Factor
Theranostics 2016; 6(1):131-141. doi:10.7150/thno.11460 This issue Cite
Research Paper
NIR-Cyanine Dye Linker: a Promising Candidate for Isochronic Fluorescence Imaging in Molecular Cancer Diagnostics and Therapy Monitoring
1. German Cancer Research Center, Department of Medical Physics in Radiology, INF 280, 69120 Heidelberg, Germany
2. German Cancer Research Center, Division of Biophysics of Macromolecules, INF 580, 69120 Heidelberg, Germany
3. University of Heidelberg, Institute of Pathology, INF 224, 69120 Heidelberg, Germany
4. German Cancer Research Center, Department of Translational Immunology, INF 580, 69120 Heidelberg, Germany
5. University of Heidelberg, Department of Radiation Oncology, INF 400, 69120 Heidelberg, Germany
#deceased
Abstract
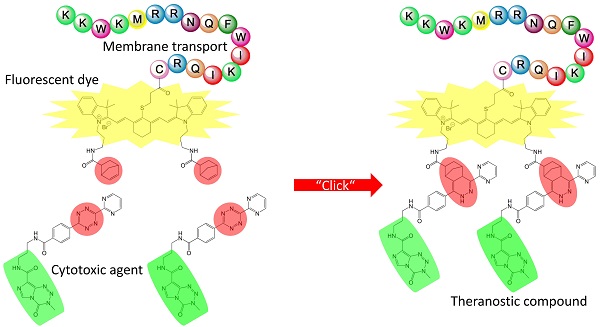
Personalized anti-cancer medicine is boosted by the recent development of molecular diagnostics and molecularly targeted drugs requiring rapid and efficient ligation routes. Here, we present a novel approach to synthetize a conjugate able to act simultaneously as an imaging and as a chemotherapeutic agent by coupling functional peptides employing solid phase peptide synthesis technologies. Development and the first synthesis of a fluorescent dye with similarity in the polymethine part of the Cy7 molecule whose indolenine-N residues were substituted with a propylene linker are described. Methylating agent temozolomide is functionalized with a tetrazine as a diene component whereas Cy7-cell penetrating peptide conjugate acts as a dienophilic reaction partner for the inverse Diels-Alder click chemistry-mediated ligation route yielding a theranostic conjugate, 3-mercapto-propionic-cyclohexenyl-Cy7-bis-temozolomide-bromide-cell penetrating peptide. Synthesis route described here may facilitate targeted delivery of the therapeutic compound to achieve sufficient local concentrations at the target site or tissue. Its versatility allows a choice of adequate imaging tags applicable in e.g. PET, SPECT, CT, near-infrared imaging, and therapeutic substances including cytotoxic agents. Imaging tags and therapeutics may be simultaneously bound to the conjugate applying click chemistry. Theranostic compound presented here offers a solid basis for a further improvement of cancer management in a precise, patient-specific manner.
Keywords: Click chemistry, inverse Diels-Alder reaction, fluorescent dye, molecular diagnostics, multimodal imaging, near infrared imaging, solid phase peptide synthesis.
 Global reach, higher impact
Global reach, higher impact