13.3
Impact Factor
Theranostics 2016; 6(2):262-271. doi:10.7150/thno.13502 This issue Cite
Research Paper
Measuring HER2-Receptor Expression In Metastatic Breast Cancer Using [68Ga]ABY-025 Affibody PET/CT
1. Section of Nuclear Medicine and PET, Department of Surgical Sciences, Uppsala University, Uppsala, Sweden.
2. Affibody AB, Solna, Sweden.
3. Department of Immunology, Genetics and Pathology, Uppsala University, Uppsala, Sweden.
4. Preclinical PET Platform, Uppsala University, Uppsala, Sweden.
5. Department of Pathology, Uppsala University Hospital, Uppsala, Sweden
Abstract
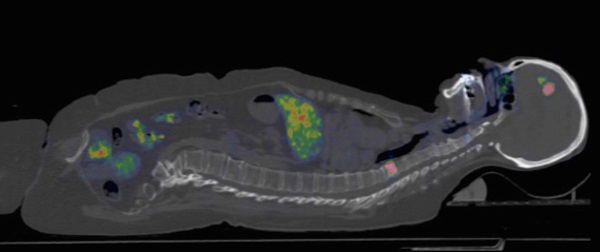
Purpose: Positron Emission Tomography (PET) imaging of HER2 expression could potentially be used to select patients for HER2-targed therapy, predict response based on uptake and be used for monitoring. In this phase I/II study the HER2-binding Affibody molecule ABY-025 was labeled with 68Ga-gallium ([68Ga]ABY-025) for PET to study effect of peptide mass, test-retest variability and correlation of quantified uptake in tumors to histopathology.
Experimental design: Sixteen women with known metastatic breast cancer and on-going treatment were included and underwent FDG PET/CT to identify viable metastases. After iv injection of 212±46 MBq [68Ga]ABY-025 whole-body PET was performed at 1, 2 and 4 h. In the first 10 patients (6 with HER2-positive and 4 with HER2-negative primary tumors), [68Ga]ABY-025 PET/CT with two different doses of injected peptide was performed one week apart. In the last six patients (5 HER2-positive and 1 HER2-negative primary tumors), repeated [68Ga]ABY-025 PET were performed one week apart as a test-retest of uptake in individual lesions. Biopsies from 16 metastases in 12 patients were collected for verification of HER2 expression by immunohistochemistry and in-situ hybridization.
Results: Imaging 4h after injection with high peptide content discriminated HER2-positive metastases best (p<0.01). PET SUV correlated with biopsy HER2-scores (r=0.91, p<0.001). Uptake was five times higher in HER2-positive than in HER2-negative lesions with no overlap (p=0.005). The test-retest intra-class correlation was r=0.996. [68Ga]ABY-025 PET correctly identified conversion and mixed expression of HER2 and targeted treatment was changed in 3 of the 16 patients.
Conclusion: [68Ga]ABY-025 PET accurately quantifies whole-body HER2-receptor status in metastatic breast cancer.
Keywords: Affibody, breast cancer metastases, clinical study, HER2-receptor, 68Ga-gallium
 Global reach, higher impact
Global reach, higher impact