Impact Factor
Theranostics 2016; 6(12):2052-2067. doi:10.7150/thno.16180 This issue Cite
Research Paper
Blocking the 4-1BB Pathway Ameliorates Crystalline Silica-induced Lung Inflammation and Fibrosis in Mice
1. Division of Pneumoconiosis, School of Public Health, China Medical University, Shenyang, PR China.
2. Department of Respiratory Medicine, Shanghai Pulmonary Hospital, Tongji University School of Medicine, Shanghai, PR China.
Abstract
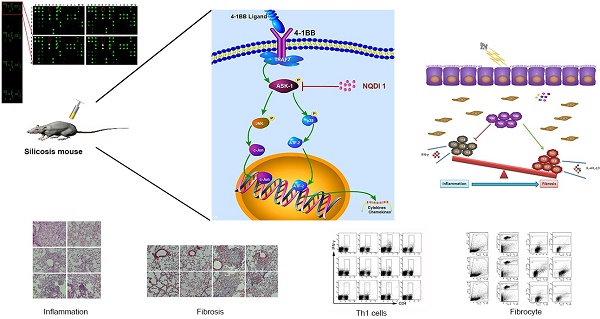
Long term pulmonary exposure to crystalline silica leads to silicosis that manifests progressive interstitial fibrosis, eventually leading to respiratory failure and death. Despite efforts to eliminate silicosis, clinical cases continue to occur in both developing and developed countries. The exact mechanisms of crystalline silica-induced pulmonary fibrosis remain elusive. Herein, we find that 4-1BB is induced in response to crystalline silica injury in lungs and that it is highly expressed during development of experimental silicosis. Therefore, we explore the role of 4-1BB pathway during crystalline silica-induced lung injury and find that a specific inhibitor blocking the pathway could effectively alleviate crystalline silica-induced lung inflammation and subsequent pulmonary fibrosis in vivo. Compared to controls, the treated mice exhibited reduced Th1 and Th17 responses. The concentrations of pro-inflammatory cytokines in bronchoalveolar lavage fluid (BALF), including tumor necrosis factor (TNF)-α, interferon (IFN)-γ and interleukin (IL)-17A following crystalline silica challenge were also reduced in inhibitor-treated mice. Although there was no significant alteration in Th2 cytokines of IL-4 and IL-13, another type of pro-fibrogenic cell, regulatory T cell (Treg) was significantly affected. In addition, one of the major participants in fibrogenesis, fibrocyte recruited less due to the blockade. Furthermore, we demonstrated the decreased fibrocyte recruitment was associated with chemokine reductions in lung. Our study discovers the 4-1BB pathway signaling enhances inflammatory response and promotes pulmonary fibrosis induced by crystalline silica. The findings here provide novel insights into the molecular events that control crystalline silica-induced lung inflammation and fibrosis through regulating Th responses and the recruitment of fibrocytes in crystalline silica-exposed lung.
Keywords: Crystalline silica, Silicosis, 4-1BB, Pulmonary fibrosis, Th responses, Fibrocyte, Molecular therapy.
 Global reach, higher impact
Global reach, higher impact