Impact Factor
Theranostics 2017; 7(2):329-343. doi:10.7150/thno.17574 This issue Cite
Research Paper
Tumor Stiffening, a Key Determinant of Tumor Progression, is Reversed by Nanomaterial-Induced Photothermal Therapy
1. Laboratoire Matière et Systèmes Complexes (MSC), CNRS-UMR 7057, Université Paris Diderot, Sorbonne Paris Cité, 10 rue Alice Domon et Léonie Duquet, F-75205 Paris Cedex 13, France
2. Plateforme Imagerie, Institut Cochin, Inserm U1016-CNRS UMR8104-Université Paris Descartes, Sorbonne Paris Cité Paris, 22 rue Méchain, 75014 Paris France
3. PARCC, Inserm U970, Paris Cardiovascular Research Center-PARCC / Université Paris-Descartes, Sorbonne Paris Cité, 56 rue Leblanc, 75015, Paris, France
4. University of Strasbourg, CNRS, Immunopathology and Therapeutic Chemistry, UPR 3572, 67000 Strasbourg, France
5. Institut Langevin, CNRS-UMR 7587, 1 place Jussieu, 75205 Paris, France.
Abstract
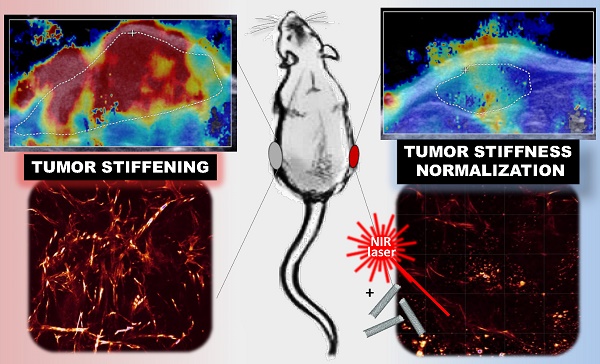
Tumor stiffening, stemming from aberrant production and organization of extracellular matrix (ECM), has been considered a predictive marker of tumor malignancy, non-invasively assessed by ultrasound shear wave elastography (SWE). Being more than a passive marker, tumor stiffening restricts the delivery of diagnostic and therapeutic agents to the tumor and per se could modulate cellular mechano-signaling, tissue inflammation and tumor progression. Current strategies to modify the tumor extracellular matrix are based on ECM-targeting chemical agents but also showed deleterious systemic effects.
On-demand excitable nanomaterials have shown their ability to perturb the tumor microenvironment in a spatiotemporal-controlled manner and synergistically with chemotherapy. Here, we investigated the evolution of tumor stiffness as well as tumor integrity and progression, under the effect of mild hyperthermia and thermal ablation generated by light-exposed multi-walled carbon nanotubes (MWCNTs) in an epidermoid carcinoma mouse xenograft. SWE was used for real-time mapping of the tumor stiffness, both during the two near infrared irradiation sessions and over the days after the treatment. We observed a transient and reversible stiffening of the tumor tissue during laser irradiation, which was lowered at the second session of mild hyperthermia or photoablation. In contrast, over the days following photothermal treatment, the treated tumors exhibited a significant softening together with volume reduction, whereas non-treated growing tumors showed an increase of tumor rigidity. The organization of the collagen matrix and the distribution of CNTs revealed a spatio-temporal correlation between the presence of nanoheaters and the damages on collagen and cells. This study highlights nanohyperthermia as a promising adjuvant strategy to reverse tumor stiffening and normalize the mechanical tumor environment.
Keywords: photothermal therapy, carbon nanotubes, cancer, elastography, tumor microenvironment.
 Global reach, higher impact
Global reach, higher impact