13.3
Impact Factor
Theranostics 2017; 7(2):390-399. doi:10.7150/thno.17087 This issue Cite
Research Paper
The Landscape of Clinical Trials Evaluating the Theranostic Role of PET Imaging in Oncology: Insights from an Analysis of ClinicalTrials.gov Database
1. Department of Radiation Oncology, Sun Yat-sen University Cancer Centre, State Key Laboratory of Oncology in South China, Collaborative Innovation Centre for Cancer Medicine, Guangzhou, People's Republic of China.
2. Clinical Trials Centre, Sun Yat-sen University Cancer Centre, State Key Laboratory of Oncology in South China, Collaborative Innovation Centre for Cancer Medicine, Guangzhou, People's Republic of China.
3. Department of Medical Statistics and Epidemiology, School of Public Health, Sun Yat-sen University, Guangzhou, People's Republic of China.
4. Department of Radiation Oncology, University of Michigan, Ann Arbor, MI, United States.
* Yu-Pei Chen, Jia-Wei Lv, and Xu Liu contributed equally to this work.
Abstract
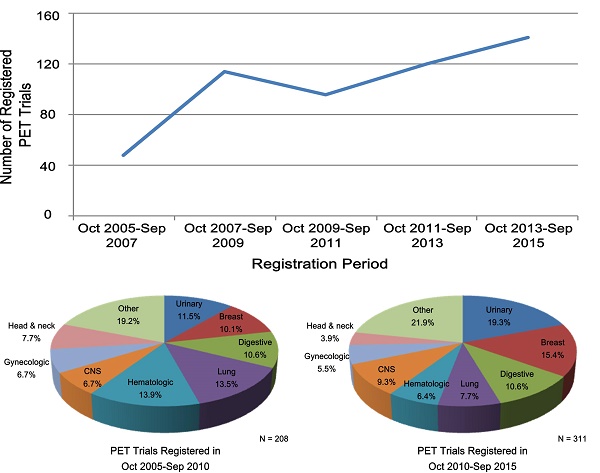
In the war on cancer marked by personalized medicine, positron emission tomography (PET)-based theranostic strategy is playing an increasingly important role. Well-designed clinical trials are of great significance for validating the PET applications and ensuring evidence-based cancer care. This study aimed to provide a comprehensive landscape of the characteristics of PET clinical trials using the substantial resource of ClinicalTrials.gov database. We identified 25,599 oncology trials registered with ClinicalTrials.gov in the last ten-year period (October 2005-September 2015). They were systematically reviewed to validate classification into 519 PET trials and 25,080 other oncology trials used for comparison. We found that PET trials were predominantly phase 1-2 studies (86.2%) and were more likely to be single-arm (78.9% vs. 57.9%, P <0.001) using non-randomized assignment (90.1% vs. 66.7%, P <0.001) than other oncology trials. Furthermore, PET trials were small in scale, generally enrolling fewer than 100 participants (20.3% vs. 25.7% for other oncology trials, P = 0.014), which might be too small to detect a significant theranostic effect. The funding support from industry or National Institutes of Health shrunk over time (both decreased by about 5%), and PET trials were more likely to be conducted in only one region lacking international collaboration (97.0% vs. 89.3% for other oncology trials, P <0.001). These findings raise concerns that clinical trials evaluating PET imaging in oncology are not receiving the attention or efforts necessary to generate high-quality evidence. Advancing the clinical application of PET imaging will require a concerted effort to improve the quality of trials.
Keywords: PET, Clinical trial, Oncology, Personalized medicine, Evidence-based care, ClinicalTrials.gov.
 Global reach, higher impact
Global reach, higher impact