13.3
Impact Factor
Theranostics 2017; 7(6):1447-1462. doi:10.7150/thno.17451 This issue Cite
Research Paper
The antihelminthic drug niclosamide effectively inhibits the malignant phenotypes of uveal melanoma in vitro and in vivo
State Key Laboratory of Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University, 54 S. Xianlie Road, Guangzhou, 510060, China.
Abstract
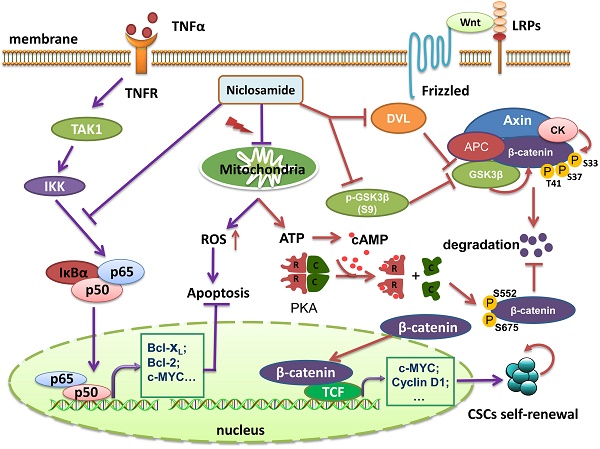
Uveal melanoma (UM) is a lethal intraocular malignancy with an average survival of only 2~8 months in patients with hepatic metastasis. Currently, there is no effective therapy for metastatic UM. Here, we reported that niclosamide, an effective repellence of tapeworm that has been approved for use in patients for approximately 50 years, exhibited strong antitumor activity in UM cells in vitro and in vivo. We showed that niclosamide potently inhibited UM cell proliferation, induced apoptosis and reduced migration and invasion. p-Niclosamide, a water-soluble niclosamide, exerted potent in vivo antitumor activity in a UM xenograft mouse model. Mechanistically, niclosamide abrogated the activation of the NF-κB pathway induced by tumor necrosis factor α (TNFα) in UM cells, while niclosamide elevated the levels of intracellullar and mitochondrial reactive oxygen species (ROS) in UM cells. Quenching ROS by N-acetylcysteine (NAC) weakened the ability of niclosamide-mediated apoptosis. Matrix metalloproteinase 9 (MMP-9) knockdown by shRNA potentiated, while ectopic expression of MMP-9 rescued, the niclosamide-attenuated invasion, implying that MMP-9 is pivotal for invasion blockage by niclosamide in UM cells. Furthermore, our results showed that niclosamide eliminated cancer stem-like cells (CSCs) as reflected by a decrease in the Aldefluor+ percentage and serial re-plating melanosphere formation, and these phenotypes were associated with the suppressed Wnt/β-catenin pathway by niclosamide in UM. Niclosamide caused a dose- and time-dependent reduction of β-catenin and the key components [e.g., DVLs, phospho-GSK3β (S9), c-Myc and Cyclin D1] in the canonical Wnt/β-catenin pathway. Additionally, niclosamide treatment in UM cells reduced ATP and cAMP contents, and decreased PKA-dependent phosphorylation of β-catenin at S552 and S675 which determine the stability of β-catenin protein, suggesting that niclosamide may work as a mitochondrial un-coupler. Taken together, our results shed light on the mechanism of antitumor action of niclosamide and warrant clinical trial for treatment of UM patients.
Keywords: niclosamide, uveal melanoma, migration, invasion, cancer stem cells
 Global reach, higher impact
Global reach, higher impact