13.3
Impact Factor
Theranostics 2017; 7(7):1976-1989. doi:10.7150/thno.17846 This issue Cite
Research Paper
Thermoacoustic Imaging and Therapy Guidance based on Ultra-short Pulsed Microwave Pumped Thermoelastic Effect Induced with Superparamagnetic Iron Oxide Nanoparticles
1. MOE Key Laboratory of Laser Life Science & Institute of Laser Life Science, College of Biophotonics, South China Normal University, Guangzhou, China;
2. Medical Imaging Center, First Affiliated Hospital of Jinan University, Guangzhou, China.
Abstract
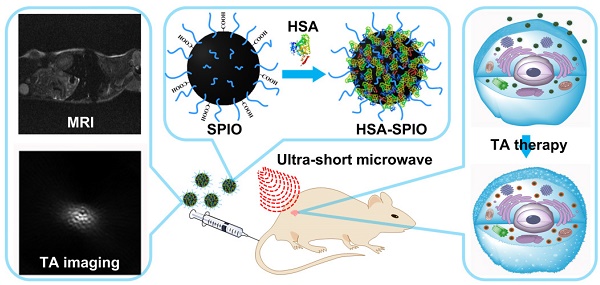
Multifunctional nanoparticle-mediated imaging and therapeutic techniques are promising modalities for accurate localization and targeted treatment of cancer in clinical settings. Thermoacoustic (TA) imaging is highly sensitive to detect the distribution of water, ions or specific nanoprobes and provides excellent resolution, good contrast and superior tissue penetrability. TA therapy is a potential non-invasive approach for the treatment of deep-seated tumors. In this study, human serum albumin (HSA)-functionalized superparamagnetic iron oxide nanoparticle (HSA-SPIO) is used as a multifunctional nanoprobe with clinical application potential for MRI, TA imaging and treatment of tumor. In addition to be a MRI contrast agent for tumor localization, HSA-SPIO can absorb pulsed microwave energy and transform it into shockwave via the thermoelastic effect. Thereby, the reconstructed TA image by detecting TA signal is expected to be a sensitive and accurate representation of the HSA-SPIO accumulation in tumor. More importantly, owing to the selective retention of HSA-SPIO in tumor tissues and strong TA shockwave at the cellular level, HSA-SPIO induced TA effect under microwave-pulse radiation can be used to highly-efficiently kill cancer cells and inhibit tumor growth. Furthermore, ultra-short pulsed microwave with high excitation efficiency and deep penetrability in biological tissues makes TA therapy a highly-efficient anti-tumor modality on the versatile platform. Overall, HSA-SPIO mediated MRI and TA imaging would offer more comprehensive diagnostic information and enable dynamic visualization of nanoagents in the tumorous tissue thereby tumor-targeted therapy.
Keywords: thermoacoustic, human serum albumin, superparamagnetic iron oxide nanoparticles, ultra-short pulsed microwave, tumor theranostic.
 Global reach, higher impact
Global reach, higher impact