Impact Factor
Theranostics 2017; 7(10):2690-2703. doi:10.7150/thno.18914 This issue Cite
Research Paper
Therapeutic Effect of Astroglia-like Mesenchymal Stem Cells Expressing Glutamate Transporter in a Genetic Rat Model of Depression
1. The Mina & Everard Goodman Faculty of Life Sciences, Bar-Ilan University, Ramat-Gan 52900, Israel;
2. Faculty of Engineering and the Institute of Nanotechnology and Advanced Materials, Bar-Ilan University, Ramat Gan 5290002, Israel;
3. Leslie Susan Gonda (Goldschmied) Multidisciplinary Brain Research Center, Bar-Ilan University, Ramat Gan 52900, Israel;
4. Hermelin Brain Tumor Center, Henry Ford Hospital, Detroit, MI, USA 48202.
Abstract
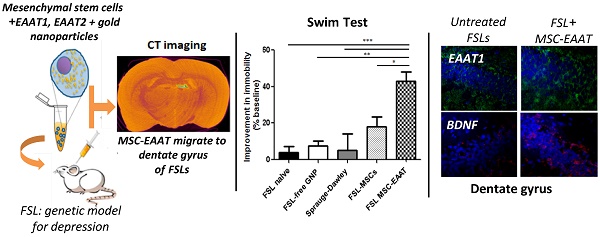
Recent studies have proposed that abnormal glutamatergic neurotransmission and glial pathology play an important role in the etiology and manifestation of depression. It was postulated that restoration of normal glutamatergic transmission, by enhancing glutamate uptake, may have a beneficial effect on depression. We examined this hypothesis using unique human glial-like mesenchymal stem cells (MSCs), which in addition to inherent properties of migration to regions of injury and secretion of neurotrophic factors, were differentiated to express high levels of functional glutamate transporters (excitatory amino acid transporters; EAAT). Additionally, gold nanoparticles (GNPs), which serve as contrast agents for CT imaging, were loaded into the cells for non-invasive, real-time imaging and tracking of MSC migration and final location within the brain. MSC-EAAT (2×105; 10 μl) were administered (i.c.v.) to Flinder Sensitive Line rats (FSLs), a genetic model for depression, and longitudinal behavioral and molecular changes were monitored. FSL rats treated with MSC-EAAT showed attenuated depressive-like behaviors (measured by the forced swim test, novelty exploration test and sucrose self-administration paradigm), as compared to controls. CT imaging, Flame Atomic Absorption Spectroscopy analysis and immunohistochemistry showed that the majority of MSCs homed specifically to the dentate gyrus of the hippocampus, a region showing structural brain changes in depression, including loss of glial cells. mRNA and protein levels of EAAT1 and BDNF were significantly elevated in the hippocampus of MSC-EAAT-treated FSLs. Our findings indicate that MSC-EAATs effectively improve depressive-like manifestations, possibly in part by increasing both glutamate uptake and neurotropic factor secretion in the hippocampus.
 Global reach, higher impact
Global reach, higher impact