Impact Factor
Theranostics 2017; 7(14):3527-3538. doi:10.7150/thno.19422 This issue Cite
Research Paper
The Role of Nitric Oxide during Sonoreperfusion of Microvascular Obstruction
1. Center for Ultrasound and Molecular Imaging and Therapeutics, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA,
2. Vascular Medicine Institute, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA.
Abstract
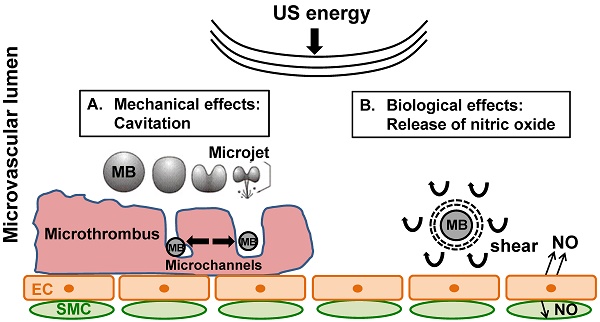
Rationale: Microembolization during PCI for acute myocardial infarction can cause microvascular obstruction (MVO). MVO severely limits the success of reperfusion therapies, is associated with additional myonecrosis, and is linked to worse prognosis, including death. We have shown, both in in vitro and in vivo models, that ultrasound (US) and microbubble (MB) therapy (termed “sonoreperfusion” or “SRP”) is a theranostic approach that relieves MVO and restores perfusion, but the underlying mechanisms remain to be established.
Objective: In this study, we investigated the role of nitric oxide (NO) during SRP.
Methods and results: We first demonstrated in plated cells that US-stimulated MB oscillations induced a 6-fold increase in endothelial nitric oxide synthase (eNOS) phosphorylation in vitro. We then monitored the kinetics of intramuscular NO and perfusion flow rate responses following 2-min of SRP therapy in the rat hindlimb muscle, with and without blockade of eNOS with LNAME. Following SRP, we found that starting at 6 minutes, intramuscular NO increased significantly over 30 min and was higher than baseline after 13 min. Concomitant contrast enhanced burst reperfusion imaging confirmed that there was a marked increase in perfusion flow rate at 6 and 10 min post SRP compared to baseline (>2.5 fold). The increases in intramuscular NO and perfusion rate were blunted with LNAME. Finally, we tested the hypothesis that NO plays a role in SRP by assessing reperfusion efficacy in a previously described rat hindlimb model of MVO during blockade of eNOS. After US treatment 1, microvascular blood volume was restored to baseline in the MB+US group, but remained low in the LNAME group. Perfusion rates increased in the MB+US group after US treatment 2 but not in the MB+US+LNAME group.
Conclusions: These data strongly support that MB oscillations can activate the eNOS pathway leading to increased blood perfusion and that NO plays a significant role in SRP efficacy.
Keywords: Microvascular obstruction, ultrasound, microbubbles, sonothrombolysis, nitric oxide
 Global reach, higher impact
Global reach, higher impact