13.3
Impact Factor
Theranostics 2017; 7(16):3989-4000. doi:10.7150/thno.21630 This issue Cite
Research Paper
Acute Inflammatory Response Following Increased Blood-Brain Barrier Permeability Induced by Focused Ultrasound is Dependent on Microbubble Dose
1. University of Toronto, Department of Medical Biophysics, Toronto, M4N 3M5, Canada;
2. Sunnybrook Research Institute, Toronto, M4N 3M5, Canada;
3. University of Toronto, Institute of Biomaterials and Biomedical Engineering, Toronto, M5S 3G9, Canada.
Abstract
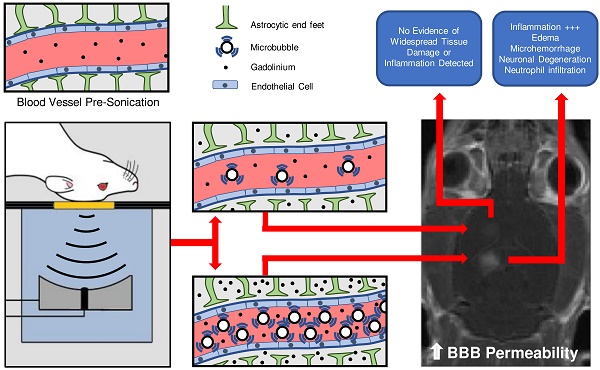
Rationale: Focused ultrasound (FUS), in conjunction with circulating microbubbles (MBs), can be used to transiently increase the permeability of the blood-brain barrier (BBB) in a targeted manner, allowing therapeutic agents to enter the brain from systemic circulation. While promising preclinical work has paved the way for the initiation of 3 human trials, there remains concern regarding neuroinflammation following treatment. The aim of this study was to assess the magnitude of this response following sonication and explore the influence of MB dose.
Methods: Differential expression of NFκB signaling pathway genes was assessed in rats at 6 h and 4 days following a FUS-mediated increase in BBB permeability. Three sonication schemes were tested: (1) a clinical imaging dose of MBs + peak negative pressure (PNP) controlled by acoustic feedback, (2) 10x clinical imaging dose of MBs + constant PNP of 0.290 MPa, and (3) 10x clinical imaging dose of MBs + PNP controlled by acoustic feedback. Follow-up magnetic resonance imaging (MRI) was performed to assess edema and hemorrhage. Hematoxylin and eosin histology was used to evaluate general tissue health.
Results: MB dose has a significant impact on the expression of several key genes involved in acute inflammation and immune activation, including Tnf, Birc3, and Ccl2. At a clinical imaging dose of MBs, there were no significant changes detected in the expression of any NFκB signaling pathway genes. Conversely, a high MB dose resulted in a clear activation of the NFκB signaling pathway, accompanied by edema, neuronal degeneration, neutrophil infiltration, and microhemorrhage. Results also suggest that post-FUS gadolinium enhancement may hold predictive value in assessing the magnitude of inflammatory response.
Conclusion: While a significant and damaging inflammatory response was observed at high MB doses, it was demonstrated that FUS can be used to induce increased BBB permeability without an associated upregulation of NFκB signaling pathway gene expression. This emphasizes the importance of employing optimized FUS parameters to mitigate the chances of causing injury to the brain at the targeted locations.
Keywords: focused ultrasound, blood-brain barrier, inflammation, microbubbles.
 Global reach, higher impact
Global reach, higher impact