13.3
Impact Factor
Theranostics 2017; 7(16):4013-4028. doi:10.7150/thno.19304 This issue Cite
Research Paper
The Use of PET Imaging for Prognostic Integrin α2β1 Phenotyping to Detect Non-Small Cell Lung Cancer and Monitor Drug Resistance Responses
1. Center for Advanced Molecular Imaging and Translation (CAMIT), Chang Gung Memorial Hospital, Tao-yuan, Taiwan;
2. Graduate Institute of Biomedical Sciences, College of Medicine, Chang Gung University, Tao-Yuan, Taiwan;
3. Department of Physiology and Pharmacology, College of Medicine, Chang Gung University, Tao-Yuan, Taiwan;
4. Molecular Medicine Research Center, College of Medicine, Chang Gung University, Tao-Yuan, Taiwan;
5. Department of Neurosurgery, Chang Gung Memorial Hospital, Tao-Yuan, Taiwan;
6. Department of Nuclear Medicine, Chang Gung Memorial Hospital, Taoyuan, Taiwan;
7. Department of Biochemical Science and Technology, College of Life Science, National Taiwan University, Taipei, Taiwan.
* Wen-Chuan Hsieh and Shih-Ting Hsu contributed equally to this work.
Abstract
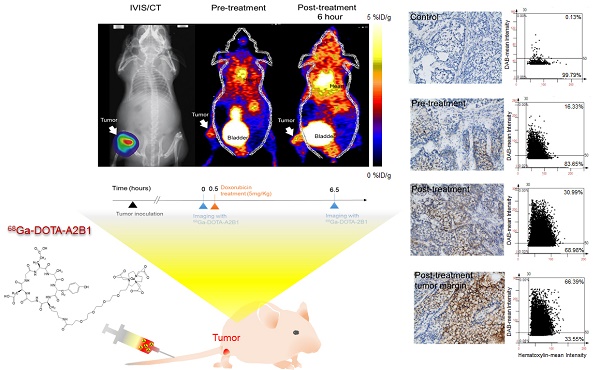
PURPOSE: Growing evidence has demonstrated that aberrant expression of integrin α2β1 might contribute to the invasion, metastasis and drug resistance of non-small cell lung cancer (NSCLC). Thus, the integrin α2β1 targeting 68Ga-DOTA-A2B1 tracer was validated in NSCLC in contrast to accumulation of the clinically used 18F-FDG PET tracer to see if 68Ga-DOTA-A2B1-PET imaging can offer a valuable and critical diagnostic imaging criterion for the identification of phenotypes of aggressive lung cancer.
METHODS: To verify the prognostic value of integrin α2β1, several quantitative and functional in vitro assays were validated in different NSCLC cell lines (CL1-0, CL1-5, A549 and selected A549++ cells). Positron emission tomography (PET) imaging studies using both standard 18F-FDG and a newly developed 68Ga-labeled integrin α2β1 (68Ga-DOTA-A2B1) tracer were sequentially performed on mice with lung tumor xenografts in different anatomic locations (subcutaneous, orthotopic and osseous) to validate the targeting capability of the 68Ga-DOTA-A2B1 tracers. Treatment responses were monitored by injecting animals with metastatic bone tumors with 5 mg/kg doxorubicin. All in vivo treatment responses in each treatment subgroup were monitored with a PET imaging system to evaluate the up-regulation of integrin expression at the earliest stage of treatment (6 h).
RESULTS: The PET and computed tomography (CT) images from NSCLC xenograft animals unambiguously demonstrated accumulation of the integrin tracer 68Ga-DOTA-A2B1 in the tumor lesions at all locations. The average tumor uptake and tumor-to-normal (T/N) ratio were 2.51 ± 0.56 %ID/g and T/N = 2.82, 3.40 ± 0.42 %ID/g and T/N = 1.52, and 1.58 ± 0.108 %ID/g and T/N = 2.31 in subcutaneous, orthotopic and osseous tumors, respectively (n = 5; p < 0.05). The xenograft tumors were all clearly visible. In contrast, the accumulation of 18F-FDG reached 3.6 ± 0.76 %ID/g, 1.39 ± 0.075 %ID/g and 3.78 ± 0.73 %ID/g in subcutaneous, orthotopic and osseous tumors, respectively (n = 5; p < 0.05). However, due to the high background uptake by normal tissue, the T/N values were less than or close to 1, making the tumors almost indistinguishable in the PET imaging analysis. Furthermore, 68Ga-DOTA-A2B1-PET imaging of the treated osseous tumor model demonstrated more than 19% tracer uptake in A549 lesions (1.72 ± 0.95 %ID/g vs. pretreatment 1.44 ± 0.12 %ID/g,p = 0. 015) 6 h post-treatment with doxorubicin. The elevated intensity of tracer uptake was in accordance with the results of in vitroWestern blot and ex vivo integrin staining, demonstrating elevated integrin α2β1 expression.
CONCLUSION: In this study, integrin α2β1 was identified as a biomarker of aggressive malignant NSCLC. Thus, efforts should be devoted to validating integrin α2β1 as a potential target for non-invasive diagnosis and as a predictive marker for monitoring treatment responses using a preclinical PET imaging system.
Keywords: Integrin α2β1, Positron emission tomography, Non-small cell lung cancer, Phenotyping imaging, Treatment response monitoring.
 Global reach, higher impact
Global reach, higher impact