13.3
Impact Factor
Theranostics 2018; 8(1):292-303. doi:10.7150/thno.22217 This issue Cite
Research Paper
Dosimetric evaluation of radionuclides for VCAM-1-targeted radionuclide therapy of early brain metastases
1. CR-UK/MRC Oxford Institute for Radiation Oncology, Department of Oncology, University of Oxford, Oxford, United Kingdom;
2. Department of Physics and Astronomy, Agnes Scott College, Decatur, GA, United States of America;
3. Departamento de Física Aplicada, Instituto de Física "Gleb Wataghin", UNICAMP, Campinas, Brazil;
4. Normandie Univ, UNICAEN, CEA, CNRS, ISTCT/CERVOxy group, Caen, France.
* Joint first authors
Abstract
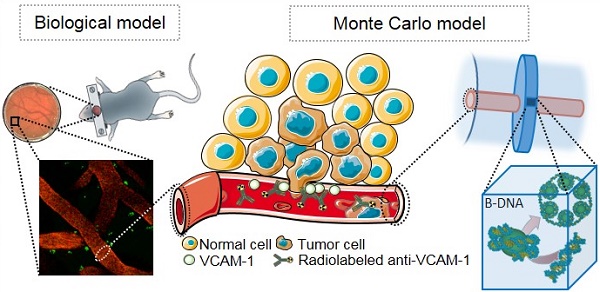
Brain metastases develop frequently in patients with breast cancer, and present a pressing therapeutic challenge. Expression of vascular cell adhesion molecule 1 (VCAM-1) is upregulated on brain endothelial cells during the early stages of metastasis and provides a target for the detection and treatment of early brain metastases. The aim of this study was to use a model of early brain metastasis to evaluate the efficacy of α-emitting radionuclides, 149Tb, 211At, 212Pb, 213Bi and 225Ac; β-emitting radionuclides, 90Y, 161Tb and 177Lu; and Auger electron (AE)-emitters 67Ga, 89Zr, 111In and 124I, for targeted radionuclide therapy (TRT).
METHODS: Histologic sections and two photon microscopy of mouse brain parenchyma were used to inform a cylindrical vessel geometry using the Geant4 general purpose Monte Carlo (MC) toolkit with the Geant4-DNA low energy physics models. Energy deposition was evaluated as a radial function and the resulting phase spaces were superimposed on a DNA model to estimate double-strand break (DSB) yields for representative β- and α-emitters, 177Lu and 212Pb. Relative biological effectiveness (RBE) values were determined by only evaluating DNA damage due to physical interactions.
RESULTS: 177Lu produced 2.69 ± 0.08 DSB per GbpGy, without significant variation from the lumen of the vessel to a radius of 100 µm. The DSB yield of 212Pb included two local maxima produced by the 6.1 MeV and 8.8 MeV α-emissions from decay products, 212Bi and 212Po, with yields of 7.64 ± 0.12 and 9.15 ± 0.24 per GbpGy, respectively. Given its higher DSB yield 212Pb may be more effective for short range targeting of early micrometastatic lesions than 177Lu.
CONCLUSION: MC simulation of a model of early brain metastases provides invaluable insight into the potential efficacy of α-, β- and AE-emitting radionuclides for TRT. 212Pb, which has the attributes of a theranostic radionuclide since it can be used for SPECT imaging, showed a favorable dose profile and RBE.
Keywords: radionuclides, VCAM-1, brain metastases
 Global reach, higher impact
Global reach, higher impact