Impact Factor
Theranostics 2018; 8(4):990-1004. doi:10.7150/thno.22465 This issue Cite
Research Paper
O2-generating MnO2 nanoparticles for enhanced photodynamic therapy of bladder cancer by ameliorating hypoxia
1. Department of Urology, Drum Tower Hospital, Medical School of Nanjing University, Institute of Urology, Nanjing University, Nanjing, Jiangsu 210008, China;
2. Department of Biomedical Engineering, College of Engineering and Applied Sciences, Collaborative Innovation Center of Chemistry for Life Sciences, Nanjing National Laboratory of Microstructures, Nanjing University, Nanjing, Jiangsu 210093, China.
* These authors contributed equally.
Abstract
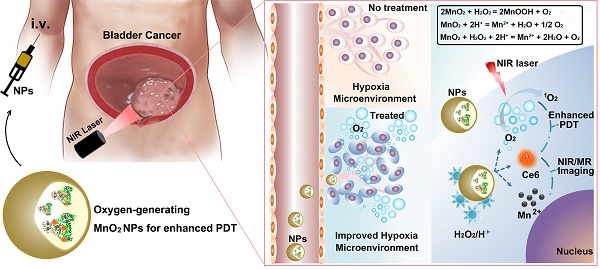
Photodynamic therapy (PDT) is an emerging effective treatment for cancer. However, the great promise of PDT for bladder cancer therapy has not yet been realized because of tumor hypoxia. To address this challenge, we fabricated O2-generating HSA-MnO2-Ce6 NPs (HSA for human serum albumin, Ce6 for chlorin e6, and NPs for nanoparticles) to overcome tumor hypoxia and thus enhance the photodynamic effect for bladder cancer therapy.
Methods: The HSA-MnO2-Ce6 NPs were prepared. We investigated the O2 generation of NPs in vitro and in vivo. The orthotopic bladder cancer model in C57BL/6 mice was established for in vivo study, and dual-modal imaging of NPs were demonstrated. Therapeutic efficacy of NPs for bladder cancer was evaluated.
Results: HSA-MnO2-Ce6 NPs had an excellent performance in generating O2 in vitro upon reaction with H2O2 at endogenous levels. Moreover, 1O2 generation was increased two-fold by using HSA-MnO2-Ce6 NPs instead of HSA-Ce6 NPs in the presence of H2O2 under 660 nm laser irradiation. In vitro cell viability assays showed that HSA-MnO2-Ce6 NPs themselves were non-toxic but greatly enhanced PDT effects on bladder cancer cells under laser irradiation. In vivo near-infrared (NIR) fluorescence and magnetic resonance (MR) imaging suggested the excellent bladder tumor-targeting property of HSA-MnO2-Ce6 NPs. O2 content in orthotopic bladder cancer was increased 3.5-fold after injection of HSA-MnO2-Ce6 NPs as compared with pre-injection. Given the excellent tumor-targeting ability and negligible toxicity, HSA-MnO2-Ce6 NPs were then used to treat orthotopic bladder cancer by PDT. The PDT with HSA-MnO2-Ce6 NPs showed remarkably improved therapeutic efficacy and significantly prolonged lifetime of mice as compared with controls.
Conclusion: This study not only demonstrated the great potential of HSA-MnO2-Ce6 NPs for bladder cancer photodynamic ablation but also provided a new therapeutic strategy to overcoming tumor hypoxia.
Keywords: enhanced photodynamic therapy, manganese dioxide (MnO2), orthotopic bladder cancer, oxygen generation, redox active nanoparticles, tumor hypoxia, tumor microenvironment.
 Global reach, higher impact
Global reach, higher impact