13.3
Impact Factor
Theranostics 2018; 8(4):1042-1058. doi:10.7150/thno.22557 This issue Cite
Research Paper
Octopod PtCu Nanoframe for Dual-Modal Imaging-Guided Synergistic Photothermal Radiotherapy
1. School of Medical Technology and Engineering, Henan University of Science and Technology, Luoyang 471023, China;
2. School of Materials Science and Engineering, Henan University of Science and Technology, Luoyang 471023, China;
3. Key Laboratory of Biorheological Science and Technology, Ministry of Education, College of Bioengineering, Chongqing University, Chongqing 400044, China.
Abstract
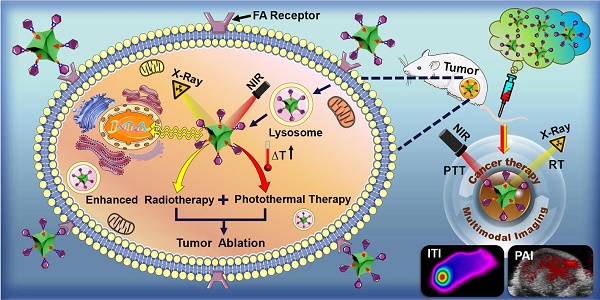
Heavy atom nanoparticles have high X-ray absorption capacity and near infrared (NIR) photothermal conversion efficiency, which could be used as radio-sensitizers. We hypothesized that concave PtCu octopod nanoframes (OPCNs) would be an efficient nanoplatform for synergistic radio-photothermal tumor ablation.
Methods: In this study, we newly exploited a folic acid-receptor (FR) mediated photothermal radiotherapy nanoagent base on OPCNs. OPCNs were synthesized with a hydrothermal method and then modified with polyethylene glycol (PEG) and folic acid (FA). A series of physical and chemical characterizations, cytotoxicity, targeting potential, endocytosis mechanism, biodistribution, systematic toxicological evaluation, pharmacokinetics, applications of OPCNs-PEG-FA for in vitro and in vivo infrared thermal imaging (ITI)/photoacoustic imaging (PAI) dual-modal imaging and synergistic photothermal radiotherapy against tumor were carried out.
Results: The OPCNs-PEG-FA demonstrated good biocompatibility, strong NIR absorption and X-ray radio-sensitization, which enabling it to track and visualize tumor in vivo via ITI/PAI dual-modal imaging. Moreover, the as-synthesized OPCNs-PEG-FA exhibited remarkable photothermal therapy (PTT) and radiotherapy (RT) synergistic tumor inhibition when treated with NIR laser and X-ray.
Conclusion: A novel multifunctional theranostic nanoplatform based on OPCNs was designed and developed for dual-modal image-guided synergistic tumor photothermal radiotherapy.
Keywords: PtCu nanoframe, targeting, near-infrared light, dual-modal imaging, synergistic tumor therapy.
 Global reach, higher impact
Global reach, higher impact