13.3
Impact Factor
Theranostics 2018; 8(4):1168-1179. doi:10.7150/thno.20271 This issue Cite
Research Paper
Diagnosis of LVAD Thrombus using a High-Avidity Fibrin-Specific 99mTc Probe
1. Division of Cardiology, Washington University School of Medicine, St. Louis, MO
2. Mallinckrodt Institute of Radiology, Washington University School of Medicine, St. Louis, MO
3. Division of Cardiothoracic Surgery, Washington University School of Medicine, St. Louis, MO
4. Barnes-Jewish Hospital, St. Louis, MO
Abstract
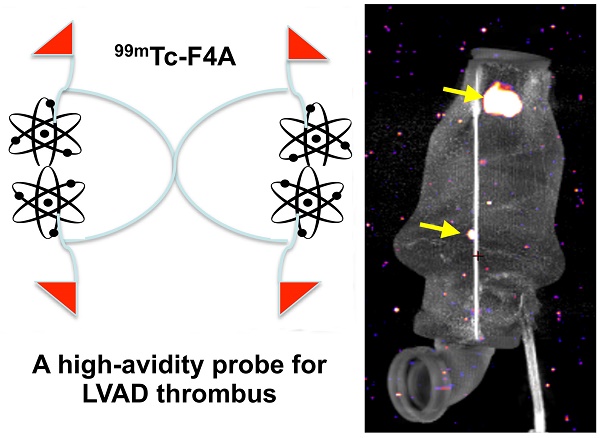
Treatment of advanced heart failure with implantable LVADs is increasing, driven by profound unmet patient need despite potential serious complications: bleeding, infection, and thrombus. The experimental objective was to develop a sensitive imaging approach to assess early thrombus accumulation in LVADs under operational high flow and high shear rates.
Methods: A monomeric bifunctional ligand with a fibrin-specific peptide, a short spacer, and 99mTc chelating amino acid sequence (F1A) was developed and compared to its tetrameric PEG analogue (F4A).
Results: 99mTc attenuation by LVAD titanium (1 mm) was 23%. 99mTc-F1A affinity to fibrin was Kd ~10 µM, whereas, the bound 99mTc-F4A probe was not displaced by F1A (120,000:1). Human plasma interfered with 99mTc-F1A binding to fibrin clot (p<0.05) in vitro, whereas, 99mTc-F4A targeting was unaffected. The pharmacokinetic half-life of 99mTc-F4A was 28% faster (124±41 min) than 99mTc-F1A (176±26 min) with both being bioeliminated through the urinary system with negligible liver or spleen biodistribution. In mice with carotid thrombus, 99mTc-F4A binding to the injured carotid was much greater (16.3±3.3 %ID/g, p=0.01) than that measured with an irrelevant negative control, 99mTc-I4A (3.4±1.6 %ID/g). In an LVAD mock flow-loop (1:1, PBS:human plasma:heparin) operating at maximal flow rate, 99mTc-F4A bound well to phantom clots in 2 min (p<0.05), whereas 99mTc-F1A had negligible targeting. Excised LVADs from patients undergoing pump exchange or heart transplant were rewired, studied in the mock flow loop, and found to have spatially variable fibrin accumulations in the inlet and outlet cannulas and bearings.
Conclusions: 99mTc-F4A is a high-avidity prototype probe for characterizing thrombus in LVADs that is anticipated to help optimize anticoagulation, reduce thromboembolic events, and minimize pump exchange.
Keywords: LVAD, thrombosis, nuclear imaging, fibrin, technetium
 Global reach, higher impact
Global reach, higher impact