13.3
Impact Factor
Theranostics 2018; 8(5):1243-1255. doi:10.7150/thno.22856 This issue Cite
Research Paper
Serum exosomes can restore cellular function in vitro and be used for diagnosis in dysferlinopathy
1. Department of Cell Biology, Tianjin Medical University, Qixiangtai Road, Heping District, Tianjin, 300070, China
2. Department of Neurology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences, Shuaifuyuan Street, Dongcheng District, Beijing, 100730, China
*These authors contribute equally to the work.
Abstract
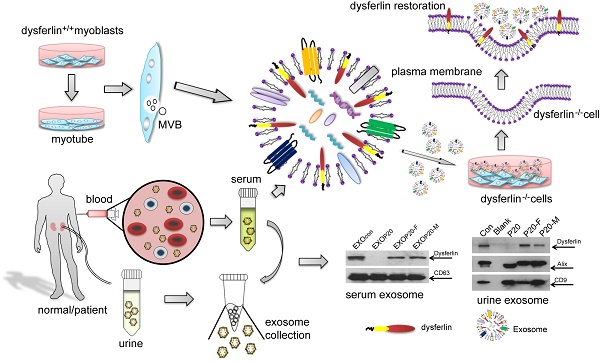
Purpose: It is challenging to deliver the full-length dysferlin gene or protein to restore cellular functions of dysferlin-deficient (DYSF-/-) myofibres in dysferlinopathy, a disease caused by the absence of dysferlin, which is currently without effective treatment. Exosomes, efficient membranous nanoscale carriers of biological cargoes, could be useful.
Experimental design: Myotube- and human serum-derived exosomes were investigated for their capabilities of restoring dysferlin protein and cellular functions in murine and human DYSF-/- cells. Moreover, dysferlinopathic patient serum- and urine-derived exosomes were assessed for their abilities as diagnostic tools for dysferlinopathy.
Results: Here we show that exosomes from dysferlin-expressing myotubes carry abundant dysferlin and enable transfer of full-length dysferlin protein to DYSF-/- myotubes. Exogenous dysferlin correctly localizes on DYSF-/- myotube membranes, enabling membrane resealing in response to injury. Human serum exosomes also carry dysferlin protein and improve membrane repair capabilities of human DYSF-/- myotubes irrespective of mutations. Lack of dysferlin in dysferlinopathic patient serum and urine exosomes enables differentiation between healthy controls and dysferlinopathic patients.
Conclusions: Our findings provide evidence that exosomes are efficient carriers of dysferlin and can be employed for the treatment and non-invasive diagnosis of dysferlinopathy.
Keywords: Dysferlinopathy, exosome, therapeutics, diagnostics
 Global reach, higher impact
Global reach, higher impact