13.3
Impact Factor
Theranostics 2018; 8(5):1327-1339. doi:10.7150/thno.21492 This issue Cite
Research Paper
Drug Release from Phase-Changeable Nanodroplets Triggered by Low-Intensity Focused Ultrasound
1. Chongqing Key Laboratory of Ultrasound Molecular Imaging, Institute of ultrasound imaging, Second Affiliated Hospital, Chongqing Medical University, Chongqing, 400010, P. R. China.
2. State Key Lab of High-Performance Ceramics and Superfine Microstructure, Shanghai Institute of Ceramics, Chinese Academy of Sciences, Shanghai, 200050, P. R. China.
Abstract
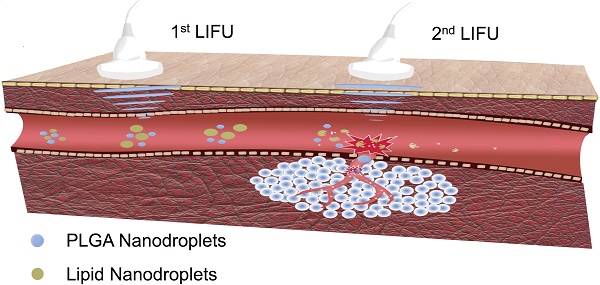
Background: As one of the most effective triggers with high tissue-penetrating capability and non-invasive feature, ultrasound shows great potential for controlling the drug release and enhancing the chemotherapeutic efficacy. In this study, we report, for the first time, construction of a phase-changeable drug-delivery nanosystem with programmable low-intensity focused ultrasound (LIFU) that could trigger drug-release and significantly enhance anticancer drug delivery.
Methods: Liquid-gas phase-changeable perfluorocarbon (perfluoropentane) and an anticancer drug (doxorubicin) were simultaneously encapsulated in two kinds of nanodroplets. By triggering LIFU, the nanodroplets could be converted into microbubbles locally in tumor tissues for acoustic imaging and the loaded anticancer drug (doxorubicin) was released after the microbubble collapse. Based on the acoustic property of shell materials, such as shell stiffness, two types of nanodroplets (lipid-based nanodroplets and PLGA-based nanodroplets) were activated by different acoustic pressure levels. Ultrasound irradiation duration and power of LIFU were tested and selected to monitor and control the drug release from nanodroplets. Various ultrasound energies were introduced to induce the phase transition and microbubble collapse of nanodroplets in vitro (3 W/3 min for lipid nanodroplets; 8 W/3 min for PLGA nanodroplets).
Results: We detected three steps in the drug-releasing profiles exhibiting the programmable patterns. Importantly, the intratumoral accumulation and distribution of the drug with LIFU exposure were significantly enhanced, and tumor proliferation was substantially inhibited. Co-delivery of two drug-loaded nanodroplets could overcome the physical barriers of tumor tissues during chemotherapy.
Conclusion: Our study provides a new strategy for the efficient ultrasound-triggered chemotherapy by nanocarriers with programmable LIFU capable of achieving the on-demand drug release.
Keywords: Programmable drug release, Low-intensity focused ultrasound (LIFU), Perfluorocarbon nanodroplets, Ultrasound imaging
 Global reach, higher impact
Global reach, higher impact