13.3
Impact Factor
Theranostics 2018; 8(5):1421-1434. doi:10.7150/thno.21906 This issue Cite
Research Paper
Selection of Tissue Factor-Deficient Cell Transplants as a Novel Strategy for Improving Hemocompatibility of Human Bone Marrow Stromal Cells
1. Spinal Cord Injury and Tissue Regeneration Center Salzburg (Sci-TReCS), Paracelsus Medical University, Salzburg, Austria;
2. Department of Transfusion Medicine, Paracelsus Medical University, Salzburg, Austria;
3. Experimental and Clinical Cell Therapy Institute, Paracelsus Medical University, Salzburg, Austria;
4. Experimental Neuroregeneration Institute, Paracelsus Medical University, Salzburg, Austria;
5. Department of Plastic, Aesthetic and Reconstructive Surgery, Hospital Barmherzige Brueder, Salzburg, Austria
* Equal contribution
Abstract
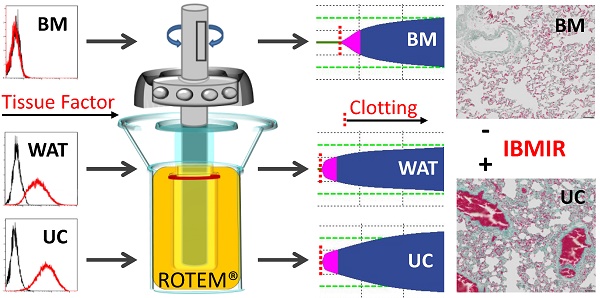
Intravascular transplantation of tissue factor (TF)-bearing cells elicits an instant blood-mediated inflammatory reaction (IBMIR) resulting in thrombotic complications and reduced engraftment. Here we studied the hemocompatibility of commonly used human white adipose tissue (WAT), umbilical cord (UC) and bone marrow stromal cells (BMSC) and devised a possible strategy for safe and efficient stromal cell transplantation.
Methods: Stromal cell identity, purity, and TF expression was tested by RTQ-PCR, flow cytometry and immunohistochemistry. Pro-coagulant activity and fibrin clot formation/stabilization was measured In Vitro by viscoelastic rotational plasma-thromboelastometry and in vivo by injecting sorted human stromal cells intravenously into rats. The impact of TF was verified in factor VII-deficient plasma and by sort-depleting TF/CD142+ BMSC.
Results: We found significantly less TF expression by a subpopulation of BMSC corresponding to reduced pro-coagulant activity. UC and WAT stroma showed broad TF expression and durable clotting. Higher cell numbers significantly increased clot formation partially dependent on coagulation factor VII. Depleting the TF/CD142+ subpopulation significantly ameliorated BMSC's hemocompatibility without affecting immunomodulation. TF-deficient BMSC did not produce thromboembolism in vivo, comparing favorably to massive intravascular thrombosis induction by TF-expressing stromal cells.
Conclusion: We demonstrate that plasma-based thromboelastometry provides a reliable tool to detect pro-coagulant activity of therapeutic cells. Selecting TF-deficient BMSC is a novel strategy for improving cell therapy applicability by reducing cell dose-dependent IBMIR risk. The particularly strong pro-coagulant activity of UC and WAT preparations sounds an additional note of caution regarding uncritical systemic application of stromal cells, particularly from non-hematopoietic extravascular sources.
Keywords: Cell transplantation, instant blood-mediated inflammatory reaction (IBMIR), tissue factor (TF), bone marrow stromal cells (BMSC), endothelial colony-forming progenitor cells (ECFC), human platelet lysate (HPL)
 Global reach, higher impact
Global reach, higher impact