13.3
Impact Factor
Theranostics 2018; 8(7):1782-1797. doi:10.7150/thno.22621 This issue Cite
Research Paper
Dual-Energy CT Imaging of Tumor Liposome Delivery After Gold Nanoparticle-Augmented Radiation Therapy
1. Center for In Vivo Microscopy, Department of Radiology, Duke University Medical Center, Durham, NC 27710, United States
2. Department of Biomedical Engineering, Duke University, Durham, NC, 27705, United States
3. Department of Pharmacology and Cancer Biology, Duke University, Durham, NC, 27705, United States
4. Department of Radiation Oncology, Duke University Medical Center, Durham, NC, 27710, United States
Abstract
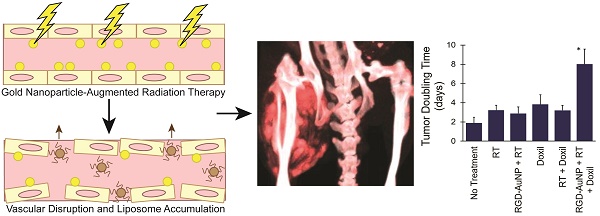
Gold nanoparticles (AuNPs) are emerging as promising agents for both cancer therapy and computed tomography (CT) imaging. AuNPs absorb x-rays and subsequently release low-energy, short-range photoelectrons during external beam radiation therapy (RT), increasing the local radiation dose. When AuNPs are near tumor vasculature, the additional radiation dose can lead to increased vascular permeability. This work focuses on understanding how tumor vascular permeability is influenced by AuNP-augmented RT, and how this effect can be used to improve the delivery of nanoparticle chemotherapeutics.
Methods: Dual-energy CT was used to quantify the accumulation of both liposomal iodine and AuNPs in tumors following AuNP-augmented RT in a mouse model of primary soft tissue sarcoma. Mice were injected with non-targeted AuNPs, RGD-functionalized AuNPs (vascular targeting), or no AuNPs, after which they were treated with varying doses of RT. The mice were injected with either liposomal iodine (for the imaging study) or liposomal doxorubicin (for the treatment study) 24 hours after RT. Increased tumor liposome accumulation was assessed by dual-energy CT (iodine) or by tracking tumor treatment response (doxorubicin).
Results: A significant increase in vascular permeability was observed for all groups after 20 Gy RT, for the targeted and non-targeted AuNP groups after 10 Gy RT, and for the vascular-targeted AuNP group after 5 Gy RT. Combining targeted AuNPs with 5 Gy RT and liposomal doxorubicin led to a significant tumor growth delay (tumor doubling time ~ 8 days) compared to AuNP-augmented RT or chemotherapy alone (tumor doubling time ~3-4 days).
Conclusions: The addition of vascular-targeted AuNPs significantly improved the treatment effect of liposomal doxorubicin after RT, consistent with the increased liposome accumulation observed in tumors in the imaging study. Using this approach with a liposomal drug delivery system can increase specific tumor delivery of chemotherapeutics, which has the potential to significantly improve tumor response and reduce the side effects of both RT and chemotherapy.
Keywords: dual-energy CT, radiation therapy, nanoparticles, tumor drug delivery, vascular imaging, small animal imaging
 Global reach, higher impact
Global reach, higher impact