13.3
Impact Factor
Theranostics 2018; 8(7):1808-1823. doi:10.7150/thno.23177 This issue Cite
Research Paper
Overcoming erlotinib resistance in EGFR mutation-positive lung adenocarcinomas through repression of phosphoglycerate dehydrogenase
1. Department of Pharmacology and Chemical Biology, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China
2. Shanghai Universities Collaborative Innovation Center for Translational Medicine, Shanghai 200025, China
3. State Key Laboratory of Oncogenes and Related Genes, Renji-Med X Clinical Stem Cell Research Center, Renji Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200127, China
4. Basic Medical College, Xinxiang Medical University, Xinxiang 453003, Henan, China
5. Department of Medicine, Cedars-Sinai Medical Center, University of California Los Angeles School of Medicine, Los Angeles, CA 90048, United States
#These authors contributed equally to the manuscript
Abstract
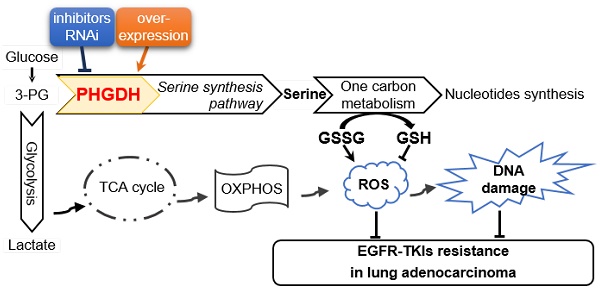
How to improve the efficacy and reverse the resistance to epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs), such as erlotinib, remains a major challenge in the targeted therapy of lung adenocarcinoma with EGFR-activating mutation. Phosphoglycerate dehydrogenase (PHGDH) is the key enzyme of de novo serine biosynthesis over-expressed in various types of cancer including lung cancer. Elevated PHGDH expression is correlated with a worse overall survival in clinical lung adenocarcinoma patients. Here we investigated the role of PHGDH in lung adenocarcinoma with the acquisition of resistance to erlotinib.
Methods: The necessary genes required for the acquired erlotinib resistance in lung adenocarcinoma cells were screened out by RNA-Seq analysis. Then the protein and mRNA levels of PHGDH were confirmed by immunoblotting and qRT-PCR in the erlotinib resistant cells. The effects of PHGDH inhibition or overexpression on erlotinib resistance were examined using cell culture and tumor xenograft mouse models respectively. To explore mechanism, the ROS level and DNA damage marker, γH2AX, were tested by DCFH-DA staining and immunofluorescence after PHGDH inhibition.
Results: We found that PHGDH level was significantly increased in the lung adenocarcinoma PC9ER4 and HCC827ER9 cells that acquired resistance to erlotinib. Perturbation of PHGDH by siPHGDH transfection or NCT-503, a small molecular PHGDH inhibitor, synergistically augmented the tumoricidal effect and restored sensitivity to erlotinib in cell lines and xenografts. Over-expression of PHGDH caused xenografts resistant to erlotinib. Furthermore, multiple DNA damage repair pathways related genes were changed by PHGDH depletion specifically in erlotinib resistant cells. ROS stress and DNA damage marker γH2AX were enhanced by siPHGDH and NCT-503, which was reversed by NAC.
Conclusion: Our study indicated that PHGDH inhibition has potential therapeutic value in lung adenocarcinoma with the acquired resistance to EGFR-TKIs.
Keywords: phosphoglycerate dehydrogenase, erlotinib resistance, metabolic activity, epidermal growth factor receptor
 Global reach, higher impact
Global reach, higher impact