Impact Factor
Theranostics 2018; 8(7):1911-1922. doi:10.7150/thno.22534 This issue Cite
Research Paper
Novel Self-assembled Organic Nanoprobe for Molecular Imaging and Treatment of Gram-positive Bacterial Infection
1. Xiangya School of Pharmaceutical Sciences, Central South University, Changsha 410013, China.
2. Hunan Provincial Research Center for Safety Evaluation of Drugs, Liuyang National Economic and Technical Development Zone 410331, China.
3. The Third Xiangya Hospital, Central South University.
Abstract
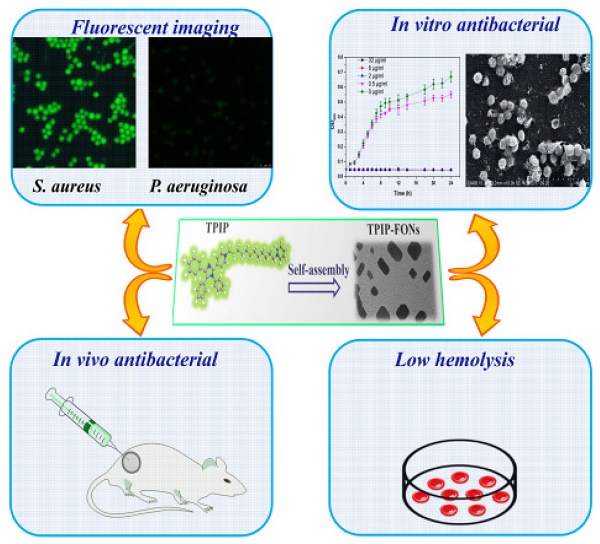
Background: Increasing bacterial infections as well as a rise in bacterial resistance call for the development of novel and safe antimicrobial agents without inducing bacterial resistance. Nanoparticles (NPs) present some advantages in treating bacterial infections and provide an alternative strategy to discover new antibiotics. Here, we report the development of novel self-assembled fluorescent organic nanoparticles (FONs) with excellent antibacterial efficacy and good biocompatibility.
Methods: Self-assembly of 1-(12-(pyridin-1-ium-1-yl)dodecyl)-4-(1,4,5-triphenyl-1H-imidazol-2-yl)pyridin-1-ium (TPIP) in aqueous solution was investigated using dynamic light scattering (DLS) and transmission electron microscopy (TEM). The bacteria were imaged under a laser scanning confocal microscope. We evaluated the antibacterial efficacy of TPIP-FONs in vitro using sugar plate test. The antimicrobial mechanism was explored by SEM. The biocompatibility of the nanoparticles was examined using cytotoxicity test, hemolysis assay, and histological staining. We further tested the antibacterial efficacy of TPIP-FONs in vivo using the S. aureus-infected rats.
Results: In aqueous solution, TPIP could self-assemble into nanoparticles (TPIP-FONs) with characteristic aggregation-induced emission (AIE). TPIP-FONs could simultaneously image gram-positive bacteria without the washing process. In vitro antimicrobial activity suggested that TPIP-FONs had excellent antibacterial activity against S. aureus (MIC = 2.0 µg mL-1). Furthermore, TPIP-FONs exhibited intrinsic biocompatibility with mammalian cells, in particular, red blood cells. In vivo studies further demonstrated that TPIP-FONs had excellent antibacterial efficacy and significantly reduced bacterial load in the infectious sites.
Conclusion: The integrated design of bacterial imaging and antibacterial functions in the self-assembled small molecules provides a promising strategy for the development of novel antimicrobial nanomaterials.
Keywords: antibacterial materials, self-assembly, aggregation-induced emission, bacterial imaging, antimicrobial activity.
 Global reach, higher impact
Global reach, higher impact