13.3
Impact Factor
Theranostics 2018; 8(7):1923-1939. doi:10.7150/thno.22834 This issue Cite
Research Paper
UTMD-Promoted Co-Delivery of Gemcitabine and miR-21 Inhibitor by Dendrimer-Entrapped Gold Nanoparticles for Pancreatic Cancer Therapy
1. Department of Ultrasound, Shanghai General Hospital, Shanghai Jiaotong University School of Medicine, Shanghai 200080, P. R. China
2. College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China
3. CQM-Centro de Química da Madeira, Universidade da Madeira, Campus da Penteada, 9000-390 Funchal, Portugal
4. Department of Instrument Science and Engineering, Shanghai Jiao Tong University, Shanghai, 201108, China
* Co-first Authors, contributed equally to this work
Abstract
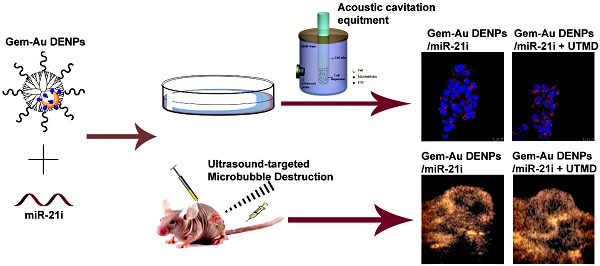
Conventional chemotherapy of pancreatic cancer (PaCa) suffers the problems of low drug permeability and inherent or acquired drug resistance. Development of new strategies for enhanced therapy still remains a great challenge. Herein, we report a new ultrasound-targeted microbubble destruction (UTMD)-promoted delivery system based on dendrimer-entrapped gold nanoparticles (Au DENPs) for co-delivery of gemcitabine (Gem) and miR-21 inhibitor (miR-21i).
Methods: In this study, Gem-Au DENPs/miR-21i was designed and synthesized. The designed polyplexes were characterized via transmission electron microscopy (TEM), Gel retardation assay and dynamic light scattering (DLS). Then, the optimum exposure parameters were examined by an ultrasound exposure platform. The cellular uptake, cytotoxicity and anticancer effects in vitro were analyzed by confocal laser microscopy, spectra microplate reader, flow cytometry and a chemiluminescence imaging system. Lastly, the anticancer effects in vivo were evaluated by contrast-enhanced ultrasound (CEUS), hematoxylin and eosin (H&E) staining, TUNEL staining and comparison of tumor volume.
Results: The results showed that the Gem-Au DENPs/miR-21i can be uptake by cancer cells and the cellular uptake was further facilitated by UTMD with an ultrasound power of 0.4 W/cm2 to enhance the cell permeability. Further, the co-delivery of Gem and miR-21i with or without UTMD treatment displayed 82-fold and 13-fold lower IC50 values than the free Gem, respectively. The UTMD-promoted co-delivery of Gem and miR-21i was further validated by in vivo treatment and showed a significant tumor volume reduction and an increase in blood perfusion of xenografted pancreatic tumors.
Conclusion: The co-delivery of Gem and miR-21i using Au DENPs can be significantly promoted by UTMD technology, hence providing a promising strategy for effective pancreatic cancer treatments.
Keywords: ultrasound-targeted microbubble destruction, PAMAM dendrimers, gold nanoparticles, drug delivery, gene delivery, pancreatic cancer
 Global reach, higher impact
Global reach, higher impact