13.3
Impact Factor
Theranostics 2018; 8(8):2134-2146. doi:10.7150/thno.22641 This issue Cite
Research Paper
Enhancement of Cancer-Specific Protoporphyrin IX Fluorescence by Targeting Oncogenic Ras/MEK Pathway
1. Division of BioMedical Sciences, Faculty of Medicine, Memorial University of Newfoundland, 300 Prince Philip Dr., St. John's, NL Canada. A1B 3V6 (709) 864-6058
2. Department of Biochemistry, Memorial University of Newfoundland, St. John's, NL Canada
*Authors contributed equally to this work.
Abstract
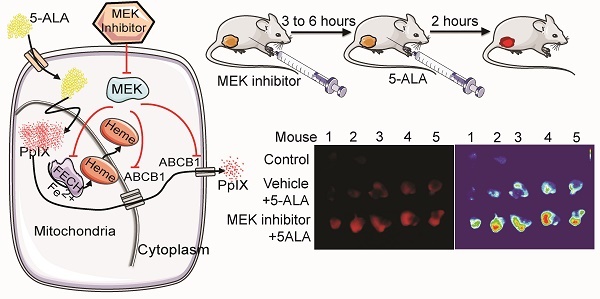
Protoporphyrin IX (PpIX) is an endogenous fluorescent molecule that selectively accumulates in cancer cells treated with the heme precursor 5-aminolevulinic acid (5-ALA). This cancer-specific accumulation of PpIX is used to distinguish tumor from normal tissues in fluorescence-guided surgery (FGS) and to destroy cancer cells by photodynamic therapy (PDT). In this study, we demonstrate that oncogenic Ras/mitogen-activated protein kinase kinase (MEK) pathway can modulate PpIX accumulation in cancer cells.
Methods: To identify Ras downstream elements involved in PpIX accumulation, chemical inhibitors were used. To demonstrate the increase of PpIX accumulation by MEK inhibition, different human normal and cancer cell lines, BALB/c mice bearing mammary 4T1 tumors and athymic nude mice bearing human tumors were used. To identify the mechanisms of PpIX regulation by MEK, biochemical and molecular biological experiments were conducted.
Results: Inhibition of one of the Ras downstream elements, MEK, promoted PpIX accumulation in cancer cells treated with 5-ALA, while inhibitors against other Ras downstream elements did not. Increased PpIX accumulation with MEK inhibition was observed in different types of human cancer cell lines, but not in normal cell lines. We identified two independent cellular mechanisms that underlie this effect in cancer cells. MEK inhibition reduced PpIX efflux from cancer cells by decreasing the expression level of ATP binding cassette subfamily B member 1 (ABCB1) transporter. In addition, the activity of ferrochelatase (FECH), the enzyme responsible for converting PpIX to heme, was reduced by MEK inhibition. Finally, we found that in vivo treatment with MEK inhibitors increased PpIX accumulation (2.2- to 2.4-fold) within mammary 4T1 tumors in BALB/c mice injected with 5-ALA without any change in normal organs. Similar results were also observed in a human tumor xenograft model.
Conclusion: Our study demonstrates that inhibition of oncogenic Ras/MEK significantly enhances PpIX accumulation in vitro and in vivo in a cancer-specific manner. Thus, suppressing the Ras/MEK pathway may be a viable strategy to selectively intensify PpIX fluorescence in cancer cells and improve its clinical applications in FGS.
Keywords: Protoporphyrin IX, fluorescence-guided surgery (FGS), Ras/MEK, ATP binding cassette subfamily B member 1 (ABCB1), ferrochelatase (FECH)
 Global reach, higher impact
Global reach, higher impact