Impact Factor
Theranostics 2018; 8(8):2161-2170. doi:10.7150/thno.23335 This issue Cite
Research Paper
Tumor-targeted Dual-modality Imaging to Improve Intraoperative Visualization of Clear Cell Renal Cell Carcinoma: A First in Man Study
1. Department of Radiology and Nuclear Medicine, Radboudumc, Nijmegen, The Netherlands.
2. Department of Urology, Radboudumc, Nijmegen, The Netherlands.
3. Department of Pathology, Radboudumc, Nijmegen, The Netherlands.
4. The Institute of Cancer Research and The Royal Marsden Hospital, London, United Kingdom.
Abstract
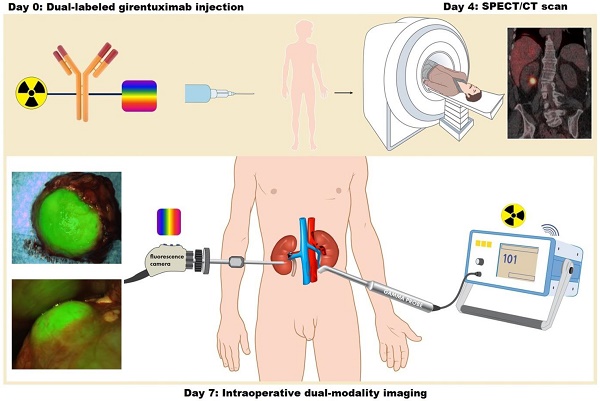
Intraoperative imaging with antibodies labeled with both a radionuclide for initial guidance and a near-infrared dye for adequate tumor delineation may overcome the main limitation of fluorescence imaging: the limited penetration depth of light in biological tissue. In this study, we demonstrate the feasibility and safety of intraoperative dual-modality imaging with the carbonic anhydrase IX (CAIX)-targeting antibody 111In-DOTA-girentuximab-IRDye800CW in clear cell renal cell carcinoma (ccRCC) patients.
Methods: A phase I protein dose escalation study was performed in patients with a primary renal mass who were scheduled for surgery. 111In-DOTA-girentuximab-IRDye800CW (5, 10, 30, or 50 mg, n=3 ccRCC patients per dose level) was administered intravenously and after 4 days SPECT/CT imaging was performed. Seven days after antibody injection, surgery was performed with the use of a gamma probe and near-infrared fluorescence camera.
Results: In total, fifteen patients were included (12 ccRCC, 3 CAIX-negative tumors). No study-related serious adverse events were observed. All ccRCC were visualized by SPECT/CT and localized by intraoperative gamma probe detection (mean tumor-to-normal kidney (T:N) ratio 2.5 ± 0.8), while the T:N ratio was 1.0 ± 0.1 in CAIX-negative tumors. ccRCC were hyperfluorescent at all protein doses and fluorescence imaging could be used for intraoperative tumor delineation, assessment of the surgical cavity and detection of (positive) surgical margins. The radiosignal was crucial for tumor localization in case of overlying fat tissue.
Conclusion: This first in man study shows that tumor-targeted dual-modality imaging using 111In-DOTA-girentuximab-IRDye800CW is safe and can be used for intraoperative guidance of ccRCC resection.
Keywords: carbonic anhydrase IX, clear cell renal cell carcinoma, intraoperative dual-modality imaging, fluorescence imaging, image-guided surgery
 Global reach, higher impact
Global reach, higher impact