13.3
Impact Factor
Theranostics 2018; 8(8):2229-2241. doi:10.7150/thno.24073 This issue Cite
Research Paper
Combining photothermal therapy and immunotherapy against melanoma by polydopamine-coated Al2O3 nanoparticles
Key Laboratory of Drug Targeting and Drug Delivery Systems, Ministry of Education, West China School of Pharmacy, Sichuan University, No.17, Block 3, Southern Renmin Road, Chengdu, 610041, P. R. China
Abstract
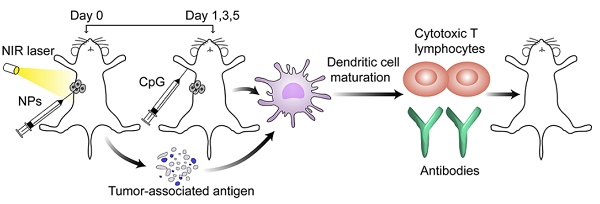
Photothermal therapy (PTT) can be an effective antitumor therapy, but it may not completely eliminate tumor cells, leading to the risk of recurrence or metastasis. Here we describe nanocarriers that allow combination therapy involving PTT and immunotherapy. Nanocarriers are prepared by coating Al2O3 nanoparticles with non-toxic, biodegradable polydopamine, which shows high photothermal efficiency. A near-infrared laser irradiation can kill the majority of tumor tissues, resulting in the release of tumor-associated antigens. The Al2O3 within the nanoparticles, together with CpG, acts as an adjuvant to trigger robust cell-mediated immune responses that can help eliminate the residual tumor cells and reduce the risk of tumor recurrence.
Methods: The characteristics and photothermal performance of polydopamine-coated Al2O3 nanoparticles were examined after one-step preparation. Then we studied their internalization, photothermal toxicity and immunostimulatory activity in vitro. For in vivo experiments, these nanocarriers were injected directly into B16F10 melanoma allografts in mice to ensure specific localization. After photothermal irradiation on day 0, mice were subcutaneously injected with CpG adjuvant on day 1, 3 and 5. Tumor volumes and number of living mice were recorded every two days. Moreover, various immune responses induced by our combined therapy were tested for mechanism research.
Results: 50% of mice after our combined treatment successfully achieved the goal of tumor eradication, and survived for 120 days, which was the end point of the experiment. Mechanism studies demonstrated the combined therapy efficiently led to dendritic cell maturation, resulting in the secretion of antibodies and cytokines as well as the proliferation of splenocytes and lymphocytes for anti-tumor immunotherapy.
Conclusion: Taken together, these results demonstrated the promise of our combined photothermal therapy and immunotherapy for tumor shrinkage, which merited further research.
Keywords: photothermal therapy, immunotherapy, Al2O3 nanoparticles, polydopamine, melanoma.
 Global reach, higher impact
Global reach, higher impact