Impact Factor
Theranostics 2018; 8(17):4649-4663. doi:10.7150/thno.26789 This issue Cite
Research Paper
Hypoxia-induced up-regulation of VASP promotes invasiveness and metastasis of hepatocellular carcinoma
Department of Hepatobiliary Surgery, the First Affiliated Hospital of Xi'an Jiaotong University, 277 Yanta West Road, Xi'an 710061, China
# Contributed equally
Abstract
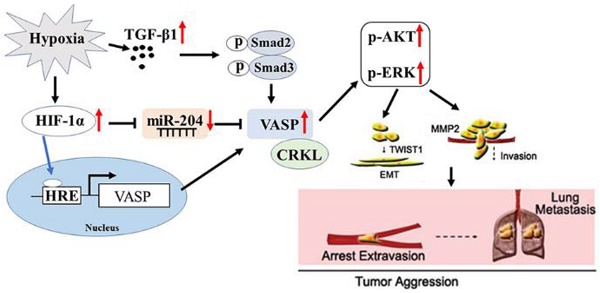
Rational: Patients with hepatocellular carcinoma (HCC) have a poor prognosis mostly due to intrahepatic as well as distal metastasis. Vasodilator-stimulated phosphoprotein (VASP), a regulator of actin cytoskeleton and cell migration, is overexpressed in HCC and correlated with its malignant features and poor prognosis. Very little is known about its function in HCC.
Methods: qRT-PCR, Western blot and IHC were used to detect the VASP expression in tissues and cells. Transwell and wound healing assays were used to measure the migration and invasion of HCC cells. Immunoblotting and immunofluorescence were used for detection of epithelial-to-mesenchymal transition (EMT) progression in HCC cells. A lung metastasis mouse model was used to evaluate metastasis of HCC in vivo. The putative targets of miR-204 were disclosed by public databases and a dual-luciferase reporter assay. IP was used to show the interaction between VASP and CRKL. ChIP was used to analyze the binding of HIF-1α to VASP promoter region.
Results: Our data involving both gain- and loss-of-function studies revealed that VASP activated AKT and ERK signaling and promoted HCC migration and invasion in vitro and in vivo by altering the EMT phenotype and expression of MMPs. We investigated the positive correlation between VASP and an adapter protein, CRKL. VASP dynamically co-localized at the SH3N domain of CRKL and mediated its function. Mechanistically, VASP overexpression at the transcriptional level was mediated by HIF-1α through direct binding to two hypoxia response elements (HRE) in the VASP promoter region. Furthermore, we identified hypoxia-induced down-regulation of miR-204, which functioned as the regulator of VASP overexpression at the post-transcriptional level. Also, hypoxia-activated p-Smad3 dependent TGF-β signaling indirectly promoted VASP expression.
Conclusion: A variety of hypoxia-induced molecular mechanisms contributed to the upregulation of VASP at transcriptional and post-transcriptional levels. These mechanisms involved CRKL, HIF-1α, miR-204, and TGF-β activating the AKT and ERK signaling to promote EMT and expression of MMPs. Taken together, our results defined VASP as an oncogene of HCC pathogenesis and metastasis with the potential to serve as a prognostic biomarker.
Keywords: VASP, hypoxia microenvironment, hepatocellular carcinoma, metastasis, CRKL
 Global reach, higher impact
Global reach, higher impact